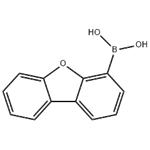
- DIBENZOFURAN-4-BORONIC ACID
-
- $50.00 / 1KG
-
2023-12-23
- CAS:100124-06-9
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: g-kg-tons, free sample is available
|
| | DIBENZOFURAN-4-BORONIC ACID Basic information |
| | DIBENZOFURAN-4-BORONIC ACID Chemical Properties |
| Melting point | 286-291 °C (lit.) | | Boiling point | 438.5±37.0 °C(Predicted) | | density | 1.34±0.1 g/cm3(Predicted) | | storage temp. | Keep in dark place,Sealed in dry,Room Temperature | | pka | 8.04±0.30(Predicted) | | form | powder | | color | white to off-white | | BRN | 1645426 | | InChIKey | ZXHUJRZYLRVVNP-UHFFFAOYSA-N | | CAS DataBase Reference | 100124-06-9(CAS DataBase Reference) |
| Hazard Codes | Xi | | Risk Statements | 36/37/38 | | Safety Statements | 37/39-26-24/25 | | WGK Germany | 3 | | TSCA | No | | HazardClass | IRRITANT | | HS Code | 29321900 |
| | DIBENZOFURAN-4-BORONIC ACID Usage And Synthesis |
| Description | Dibenzofuran-4-boronic acid (DBFBA) is a boronic acid that has been used
as a cross-coupling agent in the Suzuki coupling reaction. It has been
shown to have low efficiency, but advances in this area have been made
through the use of methanol extraction and photophysical properties.
DBFBA is often coupled with imidazole derivatives to form
anti-inflammatory compounds. The bond cleavage and hydrogen bonding
properties of DBFBA make it a good target for drug synthesis. | | Chemical Properties | off-white to beige powder | | Uses | Dibenzofuran-4-boronic acid is a water-soluble boronic acid, and the boronic acid molecule is a very useful functional group for the preparation of glycosensors. Water-soluble boronic acids that change their fluorescence properties upon binding to sugars are particularly useful as reporter units for carbohydrate fluorescence sensors.
Dibenzofuran-4-boronic acid displays distinctive fluorescence changes at three wavelengths when bound to sugars under near-physiological conditions[1]. | | Synthesis | In a 300 ml three-necked flask, 7.2 g of 4-bromodibenzofuran was dissolved in -78° C. anhydrous tetrahydrofuran (THF) under an argon atmosphere. Then, 20 ml of an n-butyl lithium-n-hexane solution (1.6 M, 1.1 eq) was added and stirred for 1 hour. 4.23 ml (1.3 eq) of trimethoxyborane (B(OMe)3) was added and stirred for 2 hours, and the temperature of the reaction system was increased to room temperature. 200 ml of 1 N hydrochloric acid was added into the reactant and stirred for 3 hours. An organic layer was separated, and solvents were distilled off. In the crude product thus obtained, hexane was added. Precipitated product was filtered to obtain 4.94 g of dibenzofuran-4-boronic acid as a white solid (yield 80%). | | References | [1] JUNFENG WANG Binghe W Shan Jin. A new boronic acid fluorescent reporter that changes emission intensities at three wavelengths upon sugar binding[J]. Tetrahedron Letters, 2005. DOI:10.1016/j.tetlet.2005.08.053. |
| | DIBENZOFURAN-4-BORONIC ACID Preparation Products And Raw materials |
|