|
|
| | (3R,4R)-1-benzyl-N,4-dimethylpiperidin-3-amine dihydrochloride Basic information |
| Product Name: | (3R,4R)-1-benzyl-N,4-dimethylpiperidin-3-amine dihydrochloride | | Synonyms: | CIS-N-BENZYL-3-METHYLAMINO-4-METHYL-PIPERIDINE BIS-(HYDROCHLORIDE);cis-1-Benzyl-N,4-dimethylpiperidin-3-amine dihydrochloride;cis-1-Benzyl-4-methyl-3-(methylamino)piperidine dihydrochloride 95+%;cis-1-Benzyl-N3,4-dimethylpiperidin-3-amine dihydrochloride;(3R,4R)-1-BENZYL-N,4-DIMETHYLPIPERIDIN-3-AMINE DIHYDROCHLORIDE;(3R,4R)-1-benzyl-N,4-diMethylpiperidin-3-aMine BIS-(HYDROCHLORIDE);3-PiperidinaMine, N,4-diMethyl-1-(phenylMethyl)-, hydrochloride (1:2), (3R,4R)-;(3R,4R)-N,4-DiMethyl-1-(phenylMethyl)-3-piperidi | | CAS: | 1062580-52-2 | | MF: | C14H23ClN2 | | MW: | 254.8 | | EINECS: | | | Product Categories: | 1 | | Mol File: | Mol File |  |
| | (3R,4R)-1-benzyl-N,4-dimethylpiperidin-3-amine dihydrochloride Chemical Properties |
| Melting point | 249~251℃ | | storage temp. | under inert gas (nitrogen or Argon) at 2-8°C | | solubility | Aqueous Acid (Slightly), DMSO (Slightly, Heated, Sonicated), Methanol (Slightly) | | form | Solid | | color | White to Off-White | | InChI | InChI=1/C14H22N2.ClH/c1-12-8-9-16(11-14(12)15-2)10-13-6-4-3-5-7-13;/h3-7,12,14-15H,8-11H2,1-2H3;1H/t12-,14+;/s3 | | InChIKey | FVRZQLQXDPJADE-WLZBWWFYNA-N | | SMILES | C(C1C=CC=CC=1)N1CC[C@@H](C)[C@@H](NC)C1.Cl |&1:10,12,r| |
| HazardClass | IRRITANT | | HS Code | 2933399990 |
| | (3R,4R)-1-benzyl-N,4-dimethylpiperidin-3-amine dihydrochloride Usage And Synthesis |
| Uses | (3R,4R)-1-benzyl-N,4-dimethylpiperidin-3-amine dihydrochloride can be used as organic synthesis intermediates and pharmaceutical intermediates, mainly used in laboratory research and development processes and chemical production processes. | | Uses | (3R,4R)-1-benzyl-N,4-dimethylpiperidin-3-amine dihydrochloride is a related compound of Tofacitinib (T528000). (3R,4R)-1-benzyl-N,4-dimethylpiperidin-3-amine dihydrochloride can be used to prepare janus kinase 1 selective inhibitor. | | Synthesis | 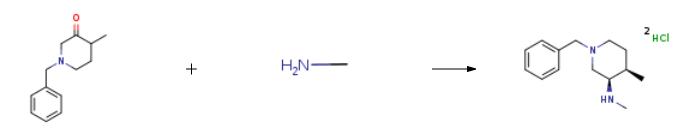
TiCl4(0.2g, 1mmol) and NEt3(0.15 g, 1.5 mmol) were added to toluene, and then add 1-benzyl-4-methyl-3-ketone piperidine (20.3 g, 100 mmol) at 35 ℃. The methylamine solution is added to the reaction solution (9.32 g, 120 mmol) and reaction for 4 h, followed by adding NaBH (OAC) 3 (0.32 g, 1.5 mmol) and glacial acetic acid 2 ml. After the reaction is complete, add saturated salt water to wash the reaction mixture. (3R,4R)-1-benzyl-N,4-dimethylpiperidin-3-amine dihydrochloride was obtained after further purification. |
| | (3R,4R)-1-benzyl-N,4-dimethylpiperidin-3-amine dihydrochloride Preparation Products And Raw materials |
|