DBCO-NHS Ester manufacturers
- DBCO-NHS ester
-
- $0.00 / 100mg
-
2024-03-20
- CAS:1353016-71-3
- Min. Order: 100mg
- Purity: >99.00%
- Supply Ability: 1g
|
| | DBCO-NHS Ester Basic information |
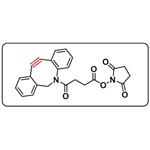
| Product Name: | DBCO-NHS Ester | | Synonyms: | DBCO-NHS;DBCO-NHS ester
(DBCO NHS ester);11,12-Didehydro-γ-oxo-dibenz[b,f]azocine-5(6H)-butanoic acid 2,5-dioxo-1-pyrrolidinyl ester;CAS_1353016-71-3;Dibenz[b,f]azocine-5(6H)-butanoic acid, 11,12-didehydro-γ-oxo-, 2,5-dioxo-1-pyrrolidinyl ester;4-[(5-aza-3,4:7,8-dibenzocyclooctan-1-yne)-5-yl]-4-oxobutyrate-n-succinimide;N-Succinimidyl 4-[(5-Aza-3,4:7,8-dibenzocyclooct-1-yne)-5-yl]-4-oxobutyrate;Dibenz[b,?f]azocine-5(6H)-butanoic acid,11,12-didehydro-γ-oxo-,2,5-dioxo-1-pyrrolidinyl ester (DBCO-NHS) | | CAS: | 1353016-71-3 | | MF: | C23H18N2O5 | | MW: | 402.4 | | EINECS: | | | Product Categories: | ADC LINKER | | Mol File: | 1353016-71-3.mol |  |
| | DBCO-NHS Ester Chemical Properties |
| Boiling point | 670.2±65.0 °C(Predicted) | | density | 1.43±0.1 g/cm3(Predicted) | | solubility | Soluble in DMSO, DCM, DMF | | pka | -0.23±0.20(Predicted) | | form | Solid | | color | White to Off-White | | Stability: | Moisture Sensitive |
| | DBCO-NHS Ester Usage And Synthesis |
| Description | DBCO-NHS Ester is a very popular amine-reactive click chemistry reagent. It can be used to modify an amine-containing molecule in organic solvents (limited solubility in aqueous media). It reacts with primary amines such as the side chain of lysine residues or aminosilane-coated surfaces at neutral or slightly basic pH to form covalent bonds. The low mass weight will add minimal spacer to modified molecules. DBCO is commonly used for copper-free Click Chemistry reactions. | | Uses | DBCO NHS ester contains an NHS carbonate group and a DBCO group. The spacer contains a photocleavable nitrobenzyloxyl group, which can be efficiently cleaved under UV to release the conjugated molecules. DBCO on the other hand readily react with azide bearing biomolecule through a copper-free click reaction instantly and in high yield.
DBCO-NHS Ester is a very popular amine-reactive click chemistry reagent. It can be used to modify an amine-containing molecule in organic solvents (limited solubility in aqueous media). It reacts with primary amines such as the side chain of lysine residues or aminosilane-coated surfaces at neutral or slightly basic pH to form covalent bonds. The low mass weight will add minimal spacer to modified molecules. DBCO is commonly used for copper-free Click Chemistry reactions. | | Uses | DBCO-NHS Ester is a derivative of DBCO (Dibenzylcyclooctyne), used in Cu-free click chemistry. It useful in strain-promoted copper-free azide-alkyne cycloaddition reactions. Used in DNA-Assisted protein analysis such as proximity ligation and extention assays. Intracellular Imaging. |
| | DBCO-NHS Ester Preparation Products And Raw materials |
| Raw materials | Dibenz[b,f]azocine-5(6H)-butanoic acid, 11,12-didehydro-γ-oxo-, methyl ester-->4-(6H-Dibenzo[b,f]azocin-5-yl)-4-oxo-butyric acid methyl ester-->Dibenz[b,f]azocine-5(6H)-butanoic acid, 11,12-dibromo-11,12-dihydro--oxo-, methyl ester-->dibenzo[b,f]azocin-6(5H)-one-->5H-dibenzo[a,d]cyclohepten-5-one oxime-->5-Dibenzosuberenone-->(Z)-5,6-dihydrodibenzo[b,f]azocine-->DBCO-Acid-->N-Hydroxysuccinimide-->N-Iodosuccinimide |
|