|
|
| | (2-((2,5-Dichloropyrimidin-4-yl)amino)phenyl)dimethylphosphine oxide Basic information |
| Product Name: | (2-((2,5-Dichloropyrimidin-4-yl)amino)phenyl)dimethylphosphine oxide | | Synonyms: | 2,5-dichloro-N-(2-(diMethylphosphoryl)phenyl)pyriMidin-4-aMine;4-(orthodimethylphosphinylanilino)-5-chloro-2-chloropyrimidine;2-[(2,5-Dichloro-4-pyrimidinyl)amino]phenyl]dimethylphosphine Oxide;2,5-Dichloro-N-[2-(dimethylphosphinyl)phenyl]-4-pyrimidinamine;4-Pyrimidinamine, 2,5-dichloro-N-[2-(dimethylphosphinyl)phenyl]-;2,5-dichloro-N-(2-(dimethylphosphoryl)phenyl)pyrimidin-4-ami...;(2-((2,5-dichloropyrimidin-4-yl)amino)phenyl)dimethylphosphine oxide;2,5- Dichloro -N-[2- (dimethylphosphino) phenyl ]-4- pyrimidinamine * | | CAS: | 1197953-49-3 | | MF: | C12H12Cl2N3OP | | MW: | 316.12 | | EINECS: | 1592732-453-0 | | Product Categories: | 1197953-49-3 | | Mol File: | 1197953-49-3.mol | 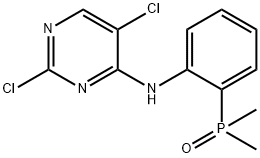 |
| | (2-((2,5-Dichloropyrimidin-4-yl)amino)phenyl)dimethylphosphine oxide Chemical Properties |
| Boiling point | 527.5±50.0 °C(Predicted) | | density | 1.41±0.1 g/cm3(Predicted) | | storage temp. | under inert gas (nitrogen or Argon) at 2-8°C | | pka | 0.30±0.10(Predicted) | | InChI | InChI=1S/C12H12Cl2N3OP/c1-19(2,18)10-6-4-3-5-9(10)16-11-8(13)7-15-12(14)17-11/h3-7H,1-2H3,(H,15,16,17) | | InChIKey | XIKUAKVCJMVXCI-UHFFFAOYSA-N | | SMILES | C1(Cl)=NC=C(Cl)C(NC2=CC=CC=C2P(C)(C)=O)=N1 |
| | (2-((2,5-Dichloropyrimidin-4-yl)amino)phenyl)dimethylphosphine oxide Usage And Synthesis |
| Uses | (2-((2,5-Dichloropyrimidin-4-yl)amino)phenyl)dimethylphosphine oxide is an intermediate of AP26113 which is a kind of effective ALK inhibitor. | | Synthesis | 25 mL of a one-necked flask was added DMF (3 mL), 2,5,6-trichloro pyrimidine (0.72 g, 3.9 mmol) was added successively with stirring,2- (dimethylphosphonino) aniline (0.5 g, 3 mmol)Anhydrous potassium carbonate (0.62 g, 4.5 mmol), heated to 60 ° C and stirred for 4 h.(30 mL x 2), the organic phase was combined, washed with water (60 mL x 2), and the organic layer was dried over anhydrous sodium sulfate, and the organic layer was washed with ethyl acetate (30 mL) and water (30 mL) Filtered and concentrated, and the residue was passed through a silica gel column to give 0.8 g of a pale yellow solid (2-((2,5-Dichloropyrimidin-4-yl)amino)phenyl)dimethylphosphine oxide.
|
| | (2-((2,5-Dichloropyrimidin-4-yl)amino)phenyl)dimethylphosphine oxide Preparation Products And Raw materials |
|