|
|
| | 4,4,4-TRICHLOROACETOACETIC ACID ETHYL ESTER Basic information |
| Product Name: | 4,4,4-TRICHLOROACETOACETIC ACID ETHYL ESTER | | Synonyms: | 4,4,4-TRICHLOROACETIC ACID ETHYL ESTER;4,4,4-TRICHLOROACETOACETIC ACID ETHYL ESTER;ETHYL 4,4,4-TRICHLOROACETOACETATE;ethyl 4,4,4-trichloro-3-oxobutyrate;4,4,4-TRICHLOROACETOACETIC ACID ETHYL ESTER, 97+%;4,4,4-trichloro-3-keto-butyric acid ethyl ester;ethyl 4,4,4-trichloro-3-oxobutanoate;ethyl 4,4,4-trichloro-3-oxo-butanoate | | CAS: | 3702-98-5 | | MF: | C6H7Cl3O3 | | MW: | 233.48 | | EINECS: | 223-041-8 | | Product Categories: | C6 to C7;Carbonyl Compounds;Esters | | Mol File: | 3702-98-5.mol | 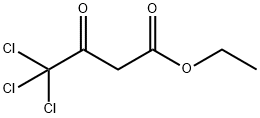 |
| | 4,4,4-TRICHLOROACETOACETIC ACID ETHYL ESTER Chemical Properties |
| Melting point | 126-127.5 °C(Solv: ligroine (8032-32-4)) | | Boiling point | 224-226 °C (lit.) | | density | 1.389 g/mL at 20 °C (lit.) | | refractive index | n20/D 1.470 | | form | clear liquid | | pka | 7.95±0.46(Predicted) | | color | Colorless to Almost colorless | | BRN | 1779520 |
| Safety Statements | 23-24/25 | | WGK Germany | 3 | | HS Code | 2918.30.9000 |
| | 4,4,4-TRICHLOROACETOACETIC ACID ETHYL ESTER Usage And Synthesis |
| Uses | Ethyl 4,4,4-Trichloro-3-oxobutanoate is a reagent used in the sunthesis of heterocyclic compounds. | | Synthesis Reference(s) | The Journal of Organic Chemistry, 31, p. 3369, 1966 DOI: 10.1021/jo01348a060 |
| | 4,4,4-TRICHLOROACETOACETIC ACID ETHYL ESTER Preparation Products And Raw materials |
|