- DDQ
-

- $0.00 / 2mg
-
2025-03-19
- CAS:84-58-2
- Min. Order: 2mg
- Purity: 99% HPLC
- Supply Ability: 20kg
|
| | 2,3-Dichloro-5,6-dicyano-1,4-benzoquinone Basic information |
| Product Name: | 2,3-Dichloro-5,6-dicyano-1,4-benzoquinone | | Synonyms: | 2,3-Dichloro-5,6-dicyano-para-benzoquinone;2-dicarbonitrile,4,5-dichloro-3,6-dioxo-4-cyclohexadiene-1;1,4-Cyclohexadiene-1,2-dicarbonitrile, 4,5-dichloro-3,6-dioxo-;2,3-DICHLORO-5,6-DICYANO-P-BENZOQUINONE ( DDQ );2,3-DICHLORO-5,6-DICYANO-P-BENZOQUINONE ( DDQ ) 99;DICHLORO(2,3-)-5,6-DICYANO-4-BENZOQUINONE;LABOTEST-BB LT00005588;1,2-DICARBONITRILE-4,5-DICHLORO-3,6-DIOXO-1,4-CYCLOHEXADIENE | | CAS: | 84-58-2 | | MF: | C8Cl2N2O2 | | MW: | 227 | | EINECS: | 201-542-2 | | Product Categories: | API intermediates;Benzoquinones;Benzoquinones, etc. (Charge Transfer Complexes);Biochemistry;Charge Transfer Complexes for Organic Metals;Functional Materials;Reagents for Oligosaccharide Synthesis;Aromatics;Intermediates;DDQ | | Mol File: | 84-58-2.mol | 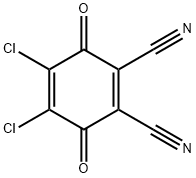 |
| | 2,3-Dichloro-5,6-dicyano-1,4-benzoquinone Chemical Properties |
| Melting point | 210-215 °C (dec.)(lit.) | | Boiling point | 301.8±42.0 °C(Predicted) | | density | 1.7500 (estimate) | | storage temp. | Store below +30°C. | | solubility | Soluble in benzene, methanol, acetic acid and dioxane.Slightly soluble in chloroform,, dichloromethane. | | form | Crystalline Powder or Crystals | | color | Yellow to orange | | Water Solubility | reacts | | Sensitive | Moisture Sensitive | | Merck | 14,3063 | | BRN | 747939 | | InChIKey | HZNVUJQVZSTENZ-UHFFFAOYSA-N | | CAS DataBase Reference | 84-58-2(CAS DataBase Reference) | | NIST Chemistry Reference | 1,4-Cyclohexadiene-1,2-dicarbonitrile, 4,5-dichloro-3,6-dioxo-(84-58-2) | | EPA Substance Registry System | 1,4-Cyclohexadiene-1,2-dicarbonitrile, 4,5-dichloro-3,6-dioxo- (84-58-2) |
| | 2,3-Dichloro-5,6-dicyano-1,4-benzoquinone Usage And Synthesis |
| Description | 2,3-Dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) has a variety of applications. DDQ is a deprotection agent for ketals, acetals, and thioacetals. It is a useful electron transfer reagent for synthesis of quinolones and an oxidizing agent used to synthesize steroids. Additionally, DDQ is used with Ph3P to synthesize 1,2-benzisoxazoles. | | Chemical Properties | yellow to orange powder | | Uses | An oxidizing agent, especially in steroid synthesis.
2,3-Dichloro-5,6-dicyano-1,4-benzoquinone is used as a reagent for oxidative couplings and cyclization reactions and dehydrogenation of alcohols, phenols and steroid ketones. It is also used in the synthesis of 1,2-benzisoxazoles. It is a useful electron-transfer reagent for synthesis of quinolines from imines and alkynes or alkenes. | | Preparation | The first synthesis of 2,3-Dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) was described by J.Thiele and F.Guntber in 1906. However, no interest was shown in the compound until Linstead and co-workers discovered its extraordinary potency as a dehydrogenating agent. Its oxidation potential is greater than that of any other known quinone.
2,3-Dichloro-5,6-Dicyanobenzoquinone (DDQ). A New Preparation | | Application | 2,3-Dichloro-5,6-dicyano-p-benzoquinone (DDQ) can be used as:
A deprotecting reagent for a variety of compounds, such as thioacetals, acetals, and ketals.
An electron-transfer reagent for the synthesis of quinolines from imines and alkynes or alkenes.
An effective reagent for the benzylic and allylic C?H functionalization.
An oxidizing agent for the synthesis of functionalized furans and benzofurans.
A reagent with Ph3P in an efficient synthesis of 1,2-benzisoxazoles. | | reaction suitability | reagent type: oxidant | | Mechanism of action | 2,3-Dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) has many applications in the oxidation of various organic compounds such as ketones, alcohols, phenols, aromatic compounds, heterocyclic structures, etc. Apart from that, the presence of two chlorine atoms and two nitrile groups on the benzoquinone ring may also behave as a potential chlorinating agent. Consequently, DDQ could act simultaneously as the chlorinating agent and the oxidant. It can also remove the protective functional groups during deprotections of different chemical entities. For dehydrogenation by DDQ, the mechanism includes the transfer of hydride to the quinone oxygen followed by the transfer of a proton to the phenolate ion[1]. | | Safety | DDQ reacts with water to release highly toxic hydrogen cyanide (HCN). A low-temperature and weakly acidic environment increases the stability of DDQ. | | Purification Methods | Crystallise DDQ from CHCl3, CHCl3/*benzene (4:1), or *benzene and store it at 0o. [Pataki & Harvey J Org Chem 52 2226 1987, Beilstein 10 H 902, 10 II 635, 10 IV 3521.] | | References | [1] Alsharif, Meshari A. et al. “DDQ as a versatile and easily recyclable oxidant: a systematic review.” RSC Advances 47 (2021): 29826–29858. |
| | 2,3-Dichloro-5,6-dicyano-1,4-benzoquinone Preparation Products And Raw materials |
|