JAK inhibitor Upadacitinib:FDA approval,Brand name,Indications and Side effects
Mar 24,2025
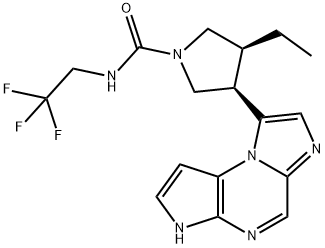
Upadacitinib is a second-generation selective Janus kinase (JAK) inhibitor targeting the JAK1 enzyme.

Upadacitinib FDA approval
Upadacitinib received FDA approval on August 16, 2019, based on positive and promising results from its multinational phase III trials in subjects with moderate to severe rheumatoid arthritis.
Upadacitinib Brand name
Rinvoq
Rinvoq LQ
Upadacitinib Indications
Upadacitinib is a new FDA-approved second-line agent for treating moderate to severe active rheumatoid arthritis (RA) in patients who have not shown an adequate response or intolerance to the first-line agent, methotrexate.
Using upadacitinib with other JAK inhibitors (JAKinibs) or robust immunosuppressants like azathioprine and cyclosporine is not advised. However, its use in combination with nonbiologic disease-modifying antirheumatic drugs (DMARDs) such as methotrexate is supported, while its use with biological DMARDs is not recommended. When combined with the first-line therapy methotrexate, upadacitinib repressed disease progression on radiographic imaging and maintained clinical efficacy.
Clinical advancements for agents used in other autoimmune diseases, such as psoriatic arthritis (PA), atopic dermatitis (AD), ankylosing spondylitis (AS), giant cell arteritis (GCA), systemic lupus erythematosus (SLE), and inflammatory bowel disease (IBD), Crohn disease (CD) and ulcerative colitis (UC), have shown promising results. Upadacitinib marks a significant milestone as the first oral medication approved by the FDA to treat moderate to severe Crohn disease.
Upadacitinib Side effects
Upadacitinib has various adverse effects, which are listed below.
Upper respiratory tract infections (URTI) (14%)
Nausea (4%)
Elevated liver enzymes (2%)
Fever (1%)
Cough (2%)
Upper respiratory tract infections (URTI) encompass:
Acute sinusitis
Laryngitis
Nasopharyngitis
Oropharyngeal pain
Pharyngitis
Pharyngotonsillitis
Rhinitis
Sinusitis
Tonsillitis
Viral upper respiratory tract infection
Upadacitinib Toxicity
Upadacitinib was found to be teratogenic in animal studies, although no human studies during pregnancy have been reported. Use during pregnancy is not recommended. Contraception is advised during treatment and for 4 weeks after completing treatment with upadacitinib.
Hepatotoxicity
The pattern of liver injury associated with upadacitinib indicates a potential for low-level, direct hepatotoxicity.
- Related articles
- Upadacitinib: Uses; Mechanism of Action; Administration and Contraindications Dec 19, 2023
Upadacitinib is FDA-approved for the treatment of moderate to severe rheumatoid arthritis (RA) that has not responded to first-line therapy.
- Synthesis and Clinical study of Upadacitinib Oct 18, 2022
Upadacitinib, a JAK1 inhibitor, has shown good efficacy in clinical trials for the treatment of a variety of autoimmune and inflammatory diseases.
Ibrutinib is a first-in-class, potent, orally administered covalently-binding inhibitor of BTK, it may stop the growth of cancer cells by blocking some of the enzymes needed for cell growth.....
Mar 25,2025APIMacitentan is an orally active, non-peptide, potent dual ERA and macitentan selectively inhibits the binding of endothelin-1 (ET-1) to ETA and ETB receptors.....
Mar 24,2025APIUpadacitinib
1310726-60-3You may like
- Upadacitinib
-

- $0.00 / 1kg
- 2025-04-03
- CAS:1310726-60-3
- Min. Order: 1kg
- Purity: 97%
- Supply Ability: 1000
- Upadacitinib
-

- $0.00 / 1Kg/Bag
- 2025-04-03
- CAS:1310726-60-3
- Min. Order: 1Kg/Bag
- Purity: 0.99
- Supply Ability: 20 tons
- *Upadacitinib
-

- $0.00 / 1g
- 2025-04-03
- CAS:1310726-60-3
- Min. Order: 1g
- Purity: 0.99
- Supply Ability: 80kg