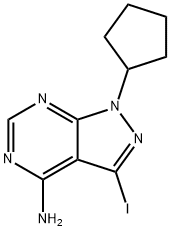
1-cyclopentyl-3-iodo-1H-pyrazolo[3,4-d]pyrimidin-4-amine synthesis
- Product Name:1-cyclopentyl-3-iodo-1H-pyrazolo[3,4-d]pyrimidin-4-amine
- CAS Number:330794-31-5
- Molecular formula:C10H12IN5
- Molecular Weight:329.14
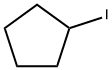
1556-18-9
89 suppliers
$38.96/5g
![3-Iodo-1H-pyrazolo[3,4-d]pyrimidin-4-amine](/CAS/GIF/151266-23-8.gif)
151266-23-8
329 suppliers
$22.00/1g
![1-cyclopentyl-3-iodo-1H-pyrazolo[3,4-d]pyrimidin-4-amine](/CAS/GIF/330794-31-5.gif)
330794-31-5
24 suppliers
inquiry
Yield:330794-31-5 60%
Reaction Conditions:
with potassium carbonate in N,N-dimethyl-formamide at 20; for 2 h;Heating / reflux;
Steps:
Synthesis of 1-cyclopentyl-3-iodo-1H-pyrazolo[3,4-d]pyrimidin-4-amine (BA80); A solution of 3-iodo-1H-pyrazolo[3,4-d]pyrimidin-4-amine (400 mg, 1.53 mmol) and K2CO3 (1 g, 6 mmol) in DMF (5 ml) was stirred at room temperature under an argon atomosphere. Iodocyclopentane (1.0 g, 0.0084 mol) was added with a syringe. Reaction was refluxed under argon atmosphere for 2 hours. Solid K2CO3 was removed by filtration. Solvent was partially removed in vacuo. Sodium citrate (50 ml) was added and reaction was extracted with EtOAc. Organic phases concentrated in vacuo and purified using silica gel column chromatography [MeOH-CH2Cl2, 5:95] yielding BA80 (300 mg, 60% yield). ESI-MS (M+H)+ m/z calcd 330.0, found 330.0.
References:
US2007/293516,2007,A1 Location in patent:Page/Page column 21
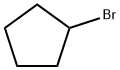
137-43-9
310 suppliers
$13.00/25g
![3-Iodo-1H-pyrazolo[3,4-d]pyrimidin-4-amine](/CAS/GIF/151266-23-8.gif)
151266-23-8
329 suppliers
$22.00/1g
![1-cyclopentyl-3-iodo-1H-pyrazolo[3,4-d]pyrimidin-4-amine](/CAS/GIF/330794-31-5.gif)
330794-31-5
24 suppliers
inquiry