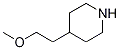
Piperidine, 4-(2-methoxyethyl)-, hydrochloride (1:1) synthesis
- Product Name:Piperidine, 4-(2-methoxyethyl)-, hydrochloride (1:1)
- CAS Number:868849-54-1
- Molecular formula:C8H17NO
- Molecular Weight:143
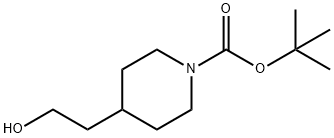
89151-44-0
226 suppliers
$8.00/1g

74-88-4
343 suppliers
$15.00/10g

868849-54-1
44 suppliers
$21.00/100mg
Yield:868849-54-1 89%
Reaction Conditions:
Stage #1: 4-(2-hydroxyethyl)piperidine-1-carboxylic acid tert-butyl esterwith sodium hydride in tetrahydrofuran at 0; for 1 h;
Stage #2: methyl iodide in tetrahydrofuran at 0 - 20;
Steps:
13.1
Intermediate 13: 1-(3-Chloro-propyl)-4-(2-methoxy-ethyl)-piperidine [] Step 1:; N-Boc-4-(2-hydroxy-ethyl)-piperidine (5g, 21.8mmol) in THF (25ml) was added to a suspension of NaH (1.47g, 43.7mmol) in THF (75ml) at 0°C under N2. The reaction was stirred for 1hour at 0°C and Mel (2.72ml, 43.7mmol) was added slowly. The reaction was allowed to warm to room temperature and stirred overnight. The reaction was quenched with water (100ml) and extracted with DCM (3 x 100ml). The combined DCM layers were dried over MgSO4, filtered, washed with DCM and concentrated in vacuo. The crude reaction (7.5g) was treated with 3M HCl solution in EtOH (150ml) for 12hours. The reaction was concentrated in vacuo and azeo-dried with toluene (2 x 150ml) to give 4-(2-methoxy-ethyl)-piperidine hydrochloride (3.5g, 89%th) as a pale yellow solid.1H NMR (400MHz, DMSO d6) δ9.25 (br s, 1 H), 9.04 (br s, 1H), 3.48 (s, 3H), 3.34 (t, J = 7Hz, 2H), 3.25-3.08 (m, 2H), 2.86-2.69 (m, 2H), 1.81-1.69 (m, 2H), 1.59 (m, 1H), 1.47-1.26 (m, 4H).
References:
EP1593679,2005,A1 Location in patent:Page/Page column 34
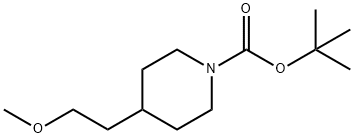
509147-79-9
11 suppliers
inquiry

868849-54-1
44 suppliers
$21.00/100mg