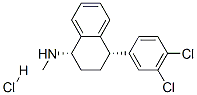
4-(3,4-Dichlorophenyl)-1,2,3,4-tetrahydro-N-methyl-1-naphthalenamine hydrochloride synthesis
- Product Name:4-(3,4-Dichlorophenyl)-1,2,3,4-tetrahydro-N-methyl-1-naphthalenamine hydrochloride
- CAS Number:79617-99-5
- Molecular formula:C17H18Cl3N
- Molecular Weight:342.69
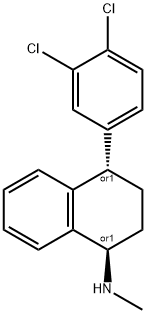
79836-45-6
23 suppliers
$329.69/10MG

79617-99-5
81 suppliers
$140.00/25mg
Yield:-
Reaction Conditions:
with hydrogenchloride in diethyl ether;tert-butyl methyl ether
Steps:
1
(IS*,4R*) formamide (1.0 g) was dissolved in THF (7 niL), and added BH3 THF (IM, 9.3 mL, 3 eq. The reaction mixture was heated to 75-80°C for 3 h and stirred at rt overnight. The reaction mixture was quenched with MeOH (20 mL). The mixture was concentrated to give a residue, which was dissolved in 10% HCl (20 mL). The solution was heated to 80-900C for 9 h, and basifed with potassium carbonate, and extracted with EtOAc (25 mL). The organic phase was separated and washed with water, brine, dried over Na2SO4. Concentrated to give the free base. It was converted to its HCl salt in TBME with HCl/Et2O to give the product (0.75g). 1H MNR (CD3OD) δ 1.86-1.96 (m, IH), 2.04-2.12 (m, IH), 2.18-2.28 (m, IH), 2.30-2.42 (m, IH), 2.78 (s, 3H), 4.34 (m, IH), 4.60 (m, IH), 6.93-7.00 (m, 2H), 7.15 (s, IH), 7.34-7.44 (m, 3H), 7.57-7.59 (d, J = 7.2 Hz, IH). Mass Spec. M+SOS.
References:
SEPRACOR INC. WO2007/6003, 2007, A2 Location in patent:Page/Page column 20; 23