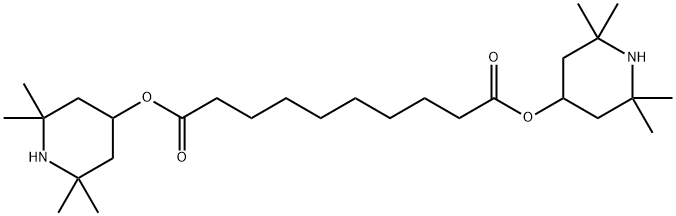
Bis(2,2,6,6-tetramethyl-4-piperidyl) sebacate synthesis
- Product Name:Bis(2,2,6,6-tetramethyl-4-piperidyl) sebacate
- CAS Number:52829-07-9
- Molecular formula:C28H52N2O4
- Molecular Weight:480.72
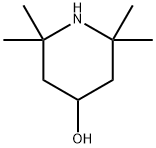
2403-88-5
339 suppliers
$5.00/10g
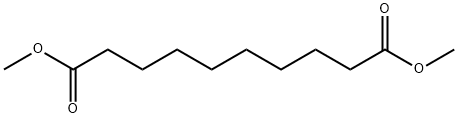
106-79-6
301 suppliers
$6.00/25g

52829-07-9
373 suppliers
$5.00/25g
Yield:-
Reaction Conditions:
with 1-methyl-pyrrolidin-2-one
Steps:
2 Example 2
Example 2 To a reaction vessel equipped with a mechanical stirrer, a thermometer, a condenser maintained at 70°C, a trap and nitrogen sparge tube are added dimethylsebacate (25 g, 109 mmoles), 2,2,6,6-tetramethyl-4-hydroxypiperidine (37.5 g, 239 mmoles) and N-methylpyrrolidinone (7 ml) and heated to 100°C. Then lithium amide (0.124 g, 5.4 mmoles) is added with agitation and the reaction mixture is heated to 150-155°C for 3 hours at atmospheric pressure. During this period, nitrogen is continuously introduced subsurface at a rate of 0.5 L/min. to remove the generated methanol. The completed reaction mass is then cooled down to 100°C and neutralized with glacial acetic acid. Analysis by gas chromatography indicated that 99% of bis(2,2,6,6-tetramethyl-4-piperidinyl)sebacate is obtained, based on the dimethylsebacate.
References:
EP510625,1992,A1

2403-88-5
339 suppliers
$5.00/10g

111-20-6
618 suppliers
$5.00/10g

52829-07-9
373 suppliers
$5.00/25g
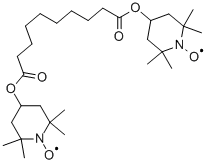
2516-92-9
107 suppliers
$105.00/1g

52829-07-9
373 suppliers
$5.00/25g
![Decanedioic acid, 1,10-bis(2,2,6,6-tetramethyl-4-piperidinyl) ester, compd. with 5,11,17,23-tetrakis(1,1-dimethylethyl)pentacyclo[19.3.1.13,7.19,13.115,19]octacosa-1(25),3,5,7(28),9,11,13(27),15,17,19(26),21,23-dodecaene-25,26,27,28-tetrol (1:2)](/CAS/20211123/GIF/138847-20-8.gif)