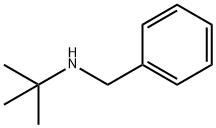
N-(tert-Butyl)benzylamine synthesis
- Product Name:N-(tert-Butyl)benzylamine
- CAS Number:3378-72-1
- Molecular formula:C11H17N
- Molecular Weight:163.26
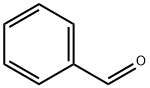
100-52-7
972 suppliers
$17.30/2g
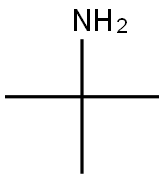
75-64-9
448 suppliers
$10.00/10g

3378-72-1
267 suppliers
$8.00/5g
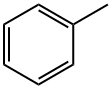
108-88-3
0 suppliers
$21.80/5 mL
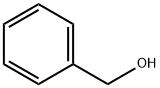
100-51-6
1434 suppliers
$5.00/100g
Yield:3378-72-1 83 %Chromat. ,108-88-3 5.5 %Chromat. ,100-51-6 8.1 %Chromat.
Reaction Conditions:
with hydrogen;5%-palladium/activated carbon at 120; under 4500.45 Torr; for 7 h;Product distribution / selectivity;Autoclave;
Steps:
3b
Examples 3 a and 3 b; In an analogous manner to that in example 1c, except using 4.0% by weight of hydrogenation catalyst, based on the total amount of amine, 1 mol of tert-butylamine was reacted with cyclohexanal (example 3a) or benzaldehyde (example 3b) to give the corresponding secondary amines (tert-butylcyclohexylmethylamine and benzyl-tert-butylamine respectively) which were then analyzed by gas chromatography. For these reactions, yields of 87.3 area % (example 3a) and 14.2 area % (example 3b) of the corresponding secondary amines were achieved.In the gas chromatography analysis for example 3a, tert-butyl cyclohexylmethyl imine (5.7 area %) was identified as the principle by-product. No detectable amounts of the corresponding tertiary amine were found. The amount of the tert-butyl cyclohexylmethyl imine by-product was reduced to a proportion of 2.3 area % by extending the continued stirring time by a further 6 h, in favor of the yield of tert-butylcyclohexylmethylamine, which rose to 90.6 area %.In example 3 b, the gas chromatography analysis found, as by-products, principally unconverted tert-butylamine (26.1 area %), benzyl alcohol (54 area %) and toluene (5.4 area %). No detectable amounts of the corresponding tertiary amine were formed. Increasing the reaction temperature to 100° C. enhanced the yield of benzyl-tert-butylamine to 19.6 area % while simultaneously reducing the formation of benzyl alcohol. Further increasing the reaction temperature to 120° C. enhanced the yield further to 83.0 area %. The by-products found at this temperature were principally unconverted tert-butylamine (3.4 area %), benzyl alcohol (8.1 area %) and toluene (5.5 area %).
References:
BASF SE US2011/251433, 2011, A1 Location in patent:Page/Page column 6
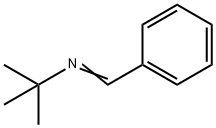
6852-58-0
46 suppliers
$28.00/5g

3378-72-1
267 suppliers
$8.00/5g

75-64-9
448 suppliers
$10.00/10g

100-51-6
1434 suppliers
$5.00/100g

3378-72-1
267 suppliers
$8.00/5g

6852-58-0
46 suppliers
$28.00/5g

3378-72-1
267 suppliers
$8.00/5g