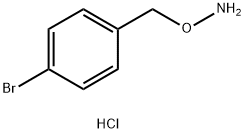
O-(4-BROMOBENZYL)HYDROXYLAMINE HYDROCHLORIDE synthesis
- Product Name:O-(4-BROMOBENZYL)HYDROXYLAMINE HYDROCHLORIDE
- CAS Number:40780-59-4
- Molecular formula:C7H9BrClNO
- Molecular Weight:238.51
![2-[(4-bromophenyl)methoxy]-2,3-dihydro-1H-isoindole-1,3-dione](/CAS/20180531/GIF/55418-33-2.gif)
55418-33-2
2 suppliers
inquiry

40780-59-4
32 suppliers
inquiry
Yield:40780-59-4 67%
Reaction Conditions:
Stage #1: 2-((4-bromobenzyl)oxy)isoindoline-1,3-dionewith hydrazine hydrate in dichloromethane at 0 - 20; for 2 h;
Stage #2: with hydrogenchloride in 1,4-dioxane at 0;
Steps:
2-9 Preparation of SAC-1109
After 595.4 mg of hydroxyphthalimide (Aldrich) was dissolved in 6 ml of dimethylformamide under argon flow, 1003.5 ml of 4-bromobenzyl bromide (Aldrich) was added, and 0.6 ml of 1,8-diazabicyclo[5.4.0]undec-7-ene (Aldrich) was slowly added. The mixture was stirred at 60° C. for 2 hours, and the temperature was lowered to room temperature. The reaction was stopped by adding a 2 N hydrochloric acid solution, followed by filtration with ethyl acetate. The solid was again dissolved in ethanol and then recrystallized, followed by filtration and drying, to obtain 932 mg of a white solid (yield: 77%). 500 mg of the obtained white solid was dissolved in 10 ml of dichloromethane, and 0.07 ml of hydrazine hydrate (Aldrich) was slowly added at 0° C. The reaction liquid was stirred at room temperature for 2 hours, and the temperature was again lowered to 0° C. The generated solid was filtered out, and the residual filtrate was concentrated under reduced pressure, followed by addition of an aqueous sodium carbonate solution and then extraction with 20 ml of diethyl ether. 3 ml of a 4 M-hydrochloric acid dioxane solution (Aldrich) was added to the solution, followed by filtration and drying, to obtain 241 mg of a solid (yield: 67%). 27 mg of the obtained solid and 50 mg of SAC-0906 obtained as obtained above were dissolved in 2 ml of pyridine (Aldrich) under argon flow, followed by stirring at room temperature for 14 hours. The reaction liquid was acidified by adding a 2 N hydrochloric acid solution, followed by extraction with 20 ml of diethyl ether, drying over magnesium sulfate, and filtration. The filtrate was concentrated under reduced pressure, and the residue was subjected to silica gel column chromatography using a mixed eluent of ethyl acetate/hexane (1:5) to obtain the target compound SAC-1109 (50 mg, yield: 75%). 1H-NMR (300 MHz, CDCl3) δ7.44-7.40 (m, 2H), 7.21-7.18 (m, 2H), 5.88-5.77 (m, 2H), 5.35-5.33 (m, 1H), 5.29-5.26 (m, 1H), 5.16 (m, 1H), 5.00 (s, 2H), 4.24-4.13 (m, 3H), 3.58-3.49 (m, 1H), 2.41-0.55 (m, 35H)
References:
US2014/378399,2014,A1 Location in patent:Paragraph 0119
![O-[(4-BROMOPHENYL)METHYL]-HYDROXYLAMINE](/CAS/GIF/55418-32-1.gif)
55418-32-1
10 suppliers
inquiry

40780-59-4
32 suppliers
inquiry
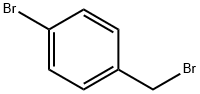
589-15-1
439 suppliers
$6.00/10g

40780-59-4
32 suppliers
inquiry
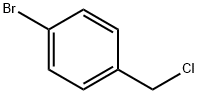
589-17-3
149 suppliers
$10.00/5g

40780-59-4
32 suppliers
inquiry