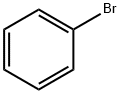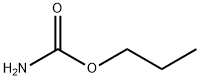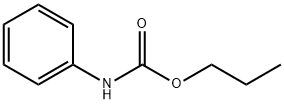
Phenylcarbamic acid propyl ester synthesis
- Product Name:Phenylcarbamic acid propyl ester
- CAS Number:5532-90-1
- Molecular formula:C10H13NO2
- Molecular Weight:179.22
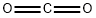
124-38-9
122 suppliers
$214.00/14L

107-08-4
246 suppliers
$14.00/5g
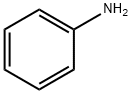
62-53-3
637 suppliers
$10.00/1g

5532-90-1
13 suppliers
inquiry
Yield:5532-90-1 94 %
Reaction Conditions:
with caesium carbonate in neat (no solvent) at 20; under 760.051 Torr;Catalytic behavior;Green chemistry;Reagent/catalyst;Solvent;
Steps:
2.3. Catalytic synthesis of carbamates from amines in CO2 pathway
General procedure: Carbamate synthesis was carried out in a 25 mL round bottom flaskfitted with a magnetic stir bar. The flask was filled with 6mmol of amine,6mmol of halide derivatives, 1.5 eq of Cs2CO3 and 10 mg of catalystZnDBPG. The RB was equipped with a balloon setup filled with CO2, 1atm. The mixture was allowed to stir for 8h. The reaction advancementwas monitored by TLC method in a regular interval. After completion,the reaction, mixture was extracted using EtOAc and work up was doneusing water and brine solution. The organic layer was separated anddried with sodium sulphate. The product conversion was measured withGC and purified with column chromatography method. All the productswere characterized using FT-IR and 1H NMR spectra.
References:
Haque, Najirul;Biswas, Surajit;Dolai, Malay;Nandi, Dip Kumar;Sarkar, Mainak;Islam, Sk Manirul [Molecular catalysis,2023,vol. 536,art. no. 112900]

682-01-9
208 suppliers
$22.00/25mL
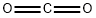
124-38-9
122 suppliers
$214.00/14L

62-53-3
637 suppliers
$10.00/1g

5532-90-1
13 suppliers
inquiry