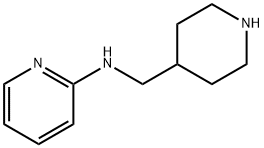
Piperidin-4-ylmethyl-pyridin-2-yl-amine synthesis
- Product Name:Piperidin-4-ylmethyl-pyridin-2-yl-amine
- CAS Number:302338-97-2
- Molecular formula:C11H17N3
- Molecular Weight:191.27
![1-Piperidinecarboxylic acid, 4-[(2-pyridinylamino)methyl]-, 1,1-dimethylethyl ester](/CAS/20210305/GIF/302341-48-6.gif)
302341-48-6
1 suppliers
inquiry

302338-97-2
23 suppliers
inquiry
Yield:302338-97-2 82%
Reaction Conditions:
with trifluoroacetic acid in dichloromethane at 20;
Steps:
39.c
1.09 g (3.74 mmoles) of (1,1-dimethylethyl) acid(4-(2-aminomethylpyridinyl)-piperidine)-1-carboxylate in 25 ml of dichloromethane with 6 ml of trifluoroacetic acid is stirred at ambient temperature until the starting product disappears according to TLC (silicagel, eluent: ethyl acetate 100). Then 20 ml of toluene is added and the mixture is evaporated to dryness under reduced pressure (2 kPa). The residue is taken up in a mixture of methylene chloride and a 2N aqueous solution of sodium hydroxide. The organic phase is decanted, washed with a saturated solution of sodium chloride, dried over magnesium sulphate and evaporated to dryness under reduced pressure (2 kPa). The oil obtained is taken up in a little diisopropyl ether and pentane then condensed to dryness under reduced pressure (2 kPa). 590 mg (Yield=82%) of expected product is obtained in the form of a yellow solid. TLC: Rf=0.2 (silicagel, eluent: dichloromethane-methanol-water-acetic acid 70-30-6-3). 1H-NMR (CDCl3): δ 1.25 and 1.8 (2m, 4H, NH-CH2--CH--CH2); 1.72 (m, 1H, CH2--CH2); 2.62 and 3.15 (2m, 6H, NH--CH2-CH-CH2- and CH--NH); 4.62 (s, 1H, mobile); 6.37 (d, 1H, CH=CH pyridine); 6.56 (m, 1H, CH=CH pyridine); 7.41 (m, 1H, CH=CH pyridine); 8.07 (m, 1H, N-=CH). MS: 192 (MH+).
References:
US2006/52398,2006,A1 Location in patent:Page/Page column 68