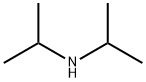
Diisopropylamine
- CAS No.
- 108-18-9
- Chemical Name:
- Diisopropylamine
- Synonyms
- DIPA;N,N-Diisopropylamine;Diisopropylamin;Diisoproplamine;Pyridine, 2-methoxy-;(iso-C3H7)2NH;AURORA KA-7634;DIISOPROPYLAMINE;Isodipropylamine;Torsemide SM3-Z2
- CBNumber:
- CB5852738
- Molecular Formula:
- C6H15N
- Molecular Weight:
- 101.19
- MDL Number:
- MFCD00008862
- MOL File:
- 108-18-9.mol
- MSDS File:
- SDS
| Melting point | -61 °C (lit.) |
|---|---|
| Boiling point | 84 °C (lit.) |
| Density | 0.722 g/mL at 25 °C (lit.) |
| vapor density | 3.5 (vs air) |
| vapor pressure | 50 mm Hg ( 20 °C) |
| refractive index |
n |
| Flash point | 1.4 °F |
| storage temp. | Store below +30°C. |
| solubility | 100 g/L (20°C) |
| pka | 11.05(at 25℃) |
| form | Liquid |
| color | Clear colorless |
| Odor | Amine. |
| PH | 11.8 (6g/l, H2O, 20℃) |
| explosive limit | 1.1-7.1%(V) |
| Water Solubility | 100 g/L (20 ºC) |
| Merck | 14,3196 |
| BRN | 605284 |
| Exposure limits | NIOSH REL: TWA 5 ppm (20 mg/m3), IDLH 200 ppm; OSHA PEL: TWA 5 ppm; ACGIH TLV: TWA 5 ppm (adopted). |
| Dielectric constant | 3.04 |
| Stability | Stable. Flammable. Incompatible with strong oxidizing agents. May react violently with strong acids or oxidizers. Air sensitive. |
| InChIKey | UAOMVDZJSHZZME-UHFFFAOYSA-N |
| LogP | 0.4 at 20℃ |
| CAS DataBase Reference | 108-18-9(CAS DataBase Reference) |
| EWG's Food Scores | 2-5 |
| FDA UNII | BR9JLI40NO |
| NIST Chemistry Reference | 2-Propanamine, N-(1-methylethyl)-(108-18-9) |
| EPA Substance Registry System | Diisopropylamine (108-18-9) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS02,GHS05,GHS06 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H225-H302-H314-H331-H335-H412 | |||||||||
| Precautionary statements | P210-P273-P280-P303+P361+P353-P304+P340+P310-P305+P351+P338 | |||||||||
| Hazard Codes | F,C | |||||||||
| Risk Statements | 11-20/22-34 | |||||||||
| Safety Statements | 16-26-36/37/39-45 | |||||||||
| OEB | A | |||||||||
| OEL | TWA: 5 ppm (20 mg/m3) [skin] | |||||||||
| RIDADR | UN 1158 3/PG 2 | |||||||||
| WGK Germany | 2 | |||||||||
| RTECS | IM4025000 | |||||||||
| Autoignition Temperature | 599 °F | |||||||||
| Hazard Note | Highly Flammable/Corrosive/Harmful | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 2921 19 99 | |||||||||
| HazardClass | 3 | |||||||||
| PackingGroup | II | |||||||||
| Toxicity | LD50 orally in rats: 0.77 g/kg (Smyth) | |||||||||
| IDLA | 200 ppm | |||||||||
| NFPA 704 |
|
Diisopropylamine price More Price(42)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | 8.03646 | Diisopropyl amine for synthesis | 108-18-9 | 10ml | $23.2 | 2024-03-01 | Buy |
| Sigma-Aldrich | 8.03646 | Diisopropyl amine for synthesis | 108-18-9 | 100mL | $35.3 | 2024-03-01 | Buy |
| Sigma-Aldrich | 8.03646 | Diisopropyl amine for synthesis | 108-18-9 | 1L | $71.1 | 2024-03-01 | Buy |
| Sigma-Aldrich | 8.03646 | Diisopropyl amine for synthesis | 108-18-9 | 2.5L | $140 | 2024-03-01 | Buy |
| Sigma-Aldrich | 8.03646 | Diisopropyl amine for synthesis | 108-18-9 | 20kg | $685 | 2024-03-01 | Buy |
Diisopropylamine Chemical Properties,Uses,Production
Description
Diisopropylamine is a secondary amine and is a colorless liquid at room temperature with a fishy, ammonia-like odor. It is used as a chemical intermediate, and catalyst for the synthesis of pesticides and pharmaceuticals. Diisopropylamine is associated with tobacco either as a natural component of tobacco, pyrolysis product (in tobacco smoke), or additive for one or more types of tobacco products. Synonyms for diisopropylamine include DIPA, and N-(1-methylethyl)-2- propanamine.
Chemical Properties
Diisopropylamine is a flammable, strongly alkaline Colorless liquid. Diisopropylamine is soluble in water and alcohol. The odor threshold ranges from 0.017 to 4.2 ppm; the fishlike odor becomes irritating at 100 mg/m3.
Physical properties
Colorless liquid with an ammonia-like odor. Experimentally determined detection and recognition odor threshold concentrations were 50 μg/m3 (130 ppbv) and 190 μg/m3 (380 ppbv), respectively (Hellman and Small, 1974).
Uses
Diisopropylamine is used as a precursor to prepare lithium diisopropylamide and sulfenamides, which are used in rubber vulcanization. It is also involved in the synthesis of N,N-Diisopropylethylamine (Hünig's base) obtained by alkylation with diethyl sulfate. Diisopropylammonium bromide is a bromide salt of DIPA used as an inert alternative to barium titanate. It is widely used as solvent as well as an intermediate in the the synthesis of pharmaceuticals, dyes, mineral flotation agents and emulsifiers.
Application
Diisopropylamine is used as a solvent and in the chemical synthesis of dyes, pharmaceuticals, and other organic syntheses. It used to synthesize diisopropylamine methyl urea (DMU), N,N-diisopropyl quinoline-2-carboxamide, 3-N-sulfonylamidine coumarins and N-chloroamine, an intermediate to generate amides.
Preparation
Diisopropylamine is produced by the reaction of diisopropyl alcohol with ammonia. Diisopropylamine is also may be prepared by the reductive amination of acetone with ammonia using a modified copper oxide, generally copper chromite, as a catalyst:
NH3+2(CH3)2CO+2H2 → C6H15N+2H2O
General Description
Diisopropylamine appears as a clear colorless liquid with an ammonia-like odor. Flash point 30°F. Less dense than water. Vapors heavier than air. Toxic oxides of nitrogen produced during combustion. Used to make other chemicals.
Air & Water Reactions
Highly flammable. Soluble in water. Sensitive to heat and air.
Reactivity Profile
Diisopropylamine can react violently with oxidizing agents and strong acids. Readily eutralizes acids in exothermic reactions to form salts plus water. May be incompatible with isocyanates, halogenated organics, peroxides, phenols (acidic), epoxides, anhydrides, and acid halides. Flammable gaseous hydrogen may be generated in combination with strong reducing agents, such as hydrides.
Health Hazard
In humans, diisopropylamine is an eye irritant. When exposed to concentrations between 25 and 50 p.p.m., workers complained of disturbances of vision. There were also complaints of nausea and headache. Dermatitis would be expected from prolonged skin exposure (Beard and Noe, 1981).
Health Hazard
Permitted levels of diisopropylamine should not cause adverse health and welfare effects. Acute inhalation exposure to diisopropylamine can cause sore throat, cough, burning sensation and shortness of breath. Adverse effects occur mainly in the upper respiratory tract, although after chronic exposure. There is no definitive evidence that diisopropylamine causes cancer so a chronic cancer value was not developed. Diisopropylamine has not been classified as causing cancer by the International Agency for Research on Cancer, the United States Environmental Protection Agency, the American Conference of Industrial Hygienists, or the National Toxicology Program.
Chemical Reactivity
Reactivity with Water No reaction; Reactivity with Common Materials: May attack some forms of plastics; Stability During Transport: Stable; Neutralizing Agents for Acids and Caustics: Not pertinent; Polymerization: Not pertinent; Inhibitor of Polymerization: Not pertinent.
Industrial uses
Diisopropylamine is used as a catalyst and as a stabilizer for mesityl oxide (HSDB 1989). It is also used as an intermediate in the synthesis of dyes, pharmaceuticals and pesticides (e.g. Diallate, Fenamiphos and Triallate).
Safety Profile
Moderately toxic by ingestion and subcutaneous routes. Mildly toxic by inhalation. Mutation data reported. A skin and severe eye irritant. Inhalation of fumes can cause pulmonary edema. A very dangerous fire hazard when exposed to heat or flame; can react vigorously with oxidizing materials. To fight fire, use alcohol foam,foam, CO2, dry chemical. When heated to decomposition it emits toxic fumes of NOx See also AMINES.
Potential Exposure
Mutagen. Thismaterial is used in the preparation of vapor-phase inhibitorsand rubber accelerators; as a chemical intermediate in thesynthesis of pharmaceuticals and pesticides (diallate, fenamiphos, and triallate, for example).
First aid
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts the skin,remove contaminated clothing and wash immediately withsoap and water. Seek medical attention immediately. If thischemical has been inhaled, remove from exposure, beginrescue breathing (using universal precautions, includingresuscitation mask) if breathing has stopped and CPR if heartaction has stopped. Transfer promptly to a medical facility.When this chemical has been swallowed, get medical attention. Give large quantities of water and induce vomiting. Donot make an unconscious person vomit. Medical observationis recommended for 24-48 h after breathing overexposure,as pulmonary edema may be delayed. As first aid for pulmonary edema, a doctor or authorized paramedic may consideradministering a corticosteroid spray.
Environmental Fate
Photolytic. Low et al. (1991) reported that the photooxidation of aqueous secondary amine
solutions by UV light in the presence of titanium dioxide resulted in the formation of ammonium
and nitrate ions.
Chemical/Physical. Reacts with acids forming water-soluble salts.
Metabolism
Little, if any investigation of the metabolism of short-chain aliphatic amines has been reported and the best that can be done is to suggest some possible metabolic routes. For example the flavin monooxygenase system could produce the hydroxylamine through N-hydroxylation catalyzed by the flavin monooxygenase system (Ziegler 1988). N-hydroxylation can also be induced with the cytochrome P-450 system as can N-dealkylation reactions (Lindeke and Cho 1982). More definitive analysis must await experimental studies.
storage
Color Code—Red: Flammability Hazard: Store ina flammable liquid storage area or approved cabinet awayfrom ignition sources and corrosive and reactive materials.Prior to working with DIPA you should be trained on itsproper handling and storage. Before entering confined spacewhere this chemical may be present, check to make surethat an explosive concentration does not exist.Diisopropylamine must be stored to avoid contact withstrong acids (such as hydrochloric, sulfuric, and nitric) oroxidizers (such as perchlorates, peroxides, permanganates,chlorates, and nitrates) because violent reactions occur.Store in tightly closed containers in a cool, well-ventilatedarea away from heat. Sources of ignition, such as smokingand open flames, are prohibited where diisopropylamine isused, handled, or stored in a manner that could create apotential fire or explosion hazard. Metal containers involving the transfer of=gallons or more of diisopropylamineshould be grounded and bonded. Drums must be equippedwith self-closing valves, pressure vacuum bungs, and flamearresters. Use only nonsparking tools and equipment, especially when opening and closing containers ofdiisopropylamine.
Shipping
This compound requires a shipping label of“FLAMMABLE LIQUID, CORROSIVE.” It falls inHazard Class 3 and Packing Group II.
Purification Methods
Distil the amine from NaOH, or reflux it three minutes over Na wire or NaH, and distil it into a dry receiver under N2. [Beilstein 4 H 154, 4 I 369, 4 II 630, 4 III 274, 4 IV 510.]
Incompatibilities
Forms explosive mixture with air. Thischemical is a strong base; reacts violently with strong oxidizers, strong acids. Attacks copper, zinc and their alloys, aluminum, and galvanized steel. Attacks some forms ofplastics and coatings.
Diisopropylamine Preparation Products And Raw materials
Raw materials
Preparation Products
1of8
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| ShanDong Look Chemical Co.,Ltd. | +8617653113219 | sales01@sdlookchemical.com | China | 2737 | 58 |
| Hebei Weibang Biotechnology Co., Ltd | +8615531157085 | abby@weibangbio.com | China | 8810 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +86 13288715578 +8613288715578 | sales@hbmojin.com | China | 12839 | 58 |
| Hebei Dangtong Import and export Co LTD | +86-13910575315 +86-13910575315 | admin@hbdangtong.com | China | 1000 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21634 | 55 |
| Shanghai Time Chemicals CO., Ltd. | +86-021-57951555 +8617317452075 | jack.li@time-chemicals.com | China | 1803 | 55 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +8618949832763 | info@tnjchem.com | China | 2986 | 55 |
| Shanxi Naipu Import and Export Co.,Ltd | +86-13734021967 +8613734021967 | kaia@neputrading.com | China | 1001 | 58 |
| career henan chemical co | +86-0371-86658258 +8613203830695 | sales@coreychem.com | China | 29881 | 58 |
| SHANDONG ZHI SHANG CHEMICAL CO.LTD | +86 18953170293 | sales@sdzschem.com | China | 2930 | 58 |
View Lastest Price from Diisopropylamine manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2024-11-27 | Diisopropylamine
108-18-9
|
US $10.00 / KG | 1KG | 99% | 10 mt | Hebei Weibang Biotechnology Co., Ltd | |
 |
2024-10-24 | Diisopropylamine
108-18-9
|
US $1.30 / kg | 10kg | 98% | 300tons | Hebei Dangtong Import and export Co LTD | |
 |
2024-10-24 | Diisopropylamine
108-18-9
|
US $50.00-40.00 / kg | 1kg | 99% | 20Tons | Hebei Dangtong Import and export Co LTD |
-

- Diisopropylamine
108-18-9
- US $10.00 / KG
- 99%
- Hebei Weibang Biotechnology Co., Ltd
-

- Diisopropylamine
108-18-9
- US $1.30 / kg
- 98%
- Hebei Dangtong Import and export Co LTD
-

- Diisopropylamine
108-18-9
- US $50.00-40.00 / kg
- 99%
- Hebei Dangtong Import and export Co LTD
108-18-9(Diisopropylamine)Related Search:
1of4