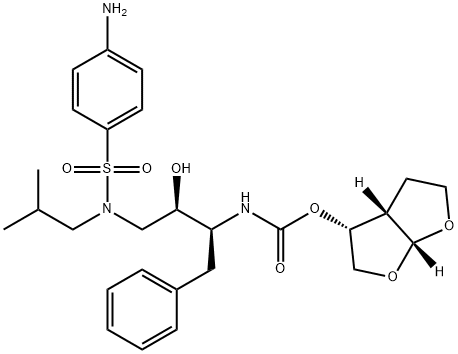
Darunavir
- CAS No.
- 206361-99-1
- Chemical Name:
- Darunavir
- Synonyms
- D03656;TMC-114;DARUVIR;arunavir;Darunavir;UIC-94017;Derunavir;Dilinavir;Dirinavir;Darunavir base
- CBNumber:
- CB51176244
- Molecular Formula:
- C27H37N3O7S
- Molecular Weight:
- 547.66
- MOL File:
- 206361-99-1.mol
- Modify Date:
- 2024/3/26 17:16:27
| Melting point | 74-760C |
|---|---|
| Density | 1.34±0.1 g/cm3(Predicted) |
| storage temp. | -20°C |
| solubility | Soluble in DMSO (>25 mg/ml) |
| pka | 11.43±0.46(Predicted) |
| form | powder |
| color | white to beige |
| Stability | Stable for 1 year from date of purchase as supplied. Solutions in DMSO may be stored at -20°C for up to 3 months. |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H302-H315-H319-H335 | |||||||||
| Precautionary statements | P261-P305+P351+P338 | |||||||||
| WGK Germany | 3 | |||||||||
| NFPA 704 |
|
Darunavir price More Price(2)
Darunavir Chemical Properties,Uses,Production
Description
Darunavir is the latest weapon in the arsenal of agents to combat human immunodeficiency virus type 1(HIV-1). As an HIV-1 protease inhibitor, its mechanism of action involves blocking the cleavage of the gag and gag–pol polyproteins into functional proteins essential for the production of infectious progeny virus; the result is the production of immature, noninfectious viral particles. Compared to predecessor HIV protease inhibitors, darunavir retains activity against resistant stains, a critical factor with the continual emergence of multidrug- resistant (MDR) mutants. Despite experiencing a 13-fold reduction in binding to MDR HIV-1 protease, this binding is 1.5 orders of magnitude tighter than the first-generation protease inhibitors. Furthermore, darunavir exhibits less than a 10-fold decrease in susceptibility in cell culture against 90% of 3309 clinical isolates resistant to amprenavir, atazanavir, indinavir, lopinavir, nelfinavir, ritonavir, saquinavir, and tipranavir. In contrast, darunavir-resistant viruses display limited susceptibility to only tipranavir, suggesting limited cross-resistance between these two protease inhibitors. To avoid the issues of the peptide-based protease inhibitors, darunavir has evolved from a structure-based design effort to minimize peptidic features and reduce molecular weight and complexity.
Chemical Properties
White Amorphous Solid
Definition
ChEBI: An N,N-disubstituted benzenesulfonamide bearing an unsubstituted amino group at the 4-position, used for the treatment of HIV infection. A second-generation HIV protease inhibitor, darunavir was designed to form robust interactions with the protease enzyme from many strains of HIV, including those from treatment-experienced patients with multiple resistance mutations to other protease inhibitors.
Acquired resistance
Darunavir is less affected than other protease inhibitors by mutations to resistance, but subgroups with more than 10 cumulative mutations show a >10-fold (median value) decrease in susceptibility. The major resistance mutations occur at positions 50 (150V), 54 (I50M/L), 76 (L76V) and 84 (I84V) of the protease gene.
Pharmaceutical Applications
A synthetic compound formulated as the ethanolate for oral use in combination with ritonavir.
Pharmacokinetics
Oral absorption: c. 82%
Cmax 600 mg once daily + ritonavir 100
mg twice daily:
c. 6500 μg/L
Cmin 600 mg oral + ritonavir
100 mg twice daily:
c. 3578 μg/L
Plasma half-life: c. 15 h
Volume of distribution: c. 131 L
Plasma protein binding: c. 95%
A single 600 mg dose given orally in combination with ritonavir
100 mg every 12 h increased the systemic exposure of
darunavir approximately 14-fold. The relative bioavailability
is 30% lower when administered with food in the presence of
low-dose ritonavir. Distribution into human CSF, semen or
breast milk has not yet been determined.
At least three oxidative metabolites, mediated predominantly
through CYP3A4, have been identified in humans;
all are at least 10-fold less active than the parent compound
against HIV. Around 80% and 14% of the dose is found in the
feces and urine, respectively. It should be used with caution
in mild–moderate hepatic impairment and avoided in patients
with more severe impairment.
Clinical Use
Treatment of HIV infection (in combination with other antiretroviral drugs)
Side effects
In phase III studies the most common adverse events were diarrhea, nausea, headache and nasopharyngitis. Patients coinfected with hepatitis B or C did not have a higher incidence of adverse events.
Darunavir Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Jigs Chemical ltd | +919099003427 | Gujarat, India | 239 | 58 | Inquiry |
| J S LABS | +91-7330612784 +91-7330612784 | Tamil Nadu, India | 160 | 58 | Inquiry |
| Varanous Labs Pvt Ltd | +91-7036248882 +91-7036248882 | Hyderabad, India | 1541 | 58 | Inquiry |
| CNS CHEMICALS | +91-9381957258 +91-9381957258 | Telangana, India | 354 | 58 | Inquiry |
| Lupin Ltd | +91-8019896181 +91-8019896181 | Maharashtra, India | 93 | 58 | Inquiry |
| Hetero Drugs Limited | +91-4023704923 +91-4023704923 | Telangana, India | 296 | 58 | Inquiry |
| HRV Global Life Sciences | +91-9820219686 +91-9820219686 | Telangana, India | 379 | 58 | Inquiry |
| Raising Sun Pharma | +91-9399941155 +91-9399941155 | Telangana, India | 127 | 58 | Inquiry |
| Laurus Labs Ltd | +91-4066594333 +91-4039804333 | Telangana, India | 50 | 58 | Inquiry |
| Arene Lifesciences Limited | +91-9849847907 +91-8455241148 | Telangana, India | 55 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| Jigs Chemical ltd | 58 |
| J S LABS | 58 |
| Varanous Labs Pvt Ltd | 58 |
| CNS CHEMICALS | 58 |
| Lupin Ltd | 58 |
| Hetero Drugs Limited | 58 |
| HRV Global Life Sciences | 58 |
| Raising Sun Pharma | 58 |
| Laurus Labs Ltd | 58 |
| Arene Lifesciences Limited | 58 |
Related articles
- Uses and Side effects of Darunavir
- Darunavir is a synthetic nonpeptidic analog of amprenavir that functions as a human immunodeficiency virus (HIV) protease inhi....
- Apr 2,2022
206361-99-1(Darunavir)Related Search:
1of4
chevron_right




