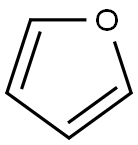
Furan
- CAS No.
- 110-00-9
- Chemical Name:
- Furan
- Synonyms
- ACETYLFURAN;ACF;1-(2-FURANYL)ETHANONE;Pyrane;tetrol;Oxole;FEMA 3163;FURYL METHYL KETONE;OXOL;axole
- CBNumber:
- CB9852796
- Molecular Formula:
- C4H4O
- Molecular Weight:
- 68.07
- MOL File:
- 110-00-9.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/6/25 17:23:33
| Melting point | -85.6 °C |
|---|---|
| Boiling point | 67 °C10 mm Hg(lit.) |
| Density | 0.936 g/mL at 25 °C(lit.) |
| vapor density | 2.35 (vs air) |
| vapor pressure | 1672 mm Hg ( 55 °C) |
| refractive index |
n |
| Flash point | 160 °F |
| storage temp. | Store below +30°C. |
| solubility | alcohols: freely soluble |
| form | Liquid |
| color | Clear colorless to yellow |
| Specific Gravity | 0.94 |
| Odor | Peculiar spicy -smokey, slightly Cinnamon-like odor |
| Odor Threshold | 9.9ppm |
| explosive limit | 2.3-14.3%(V) |
| Water Solubility | insoluble |
| Sensitive | Air & Light Sensitive |
| Merck | 14,4296 |
| BRN | 103221 |
| Dielectric constant | 3.0(25℃) |
| Exposure limits | NIOSH: IDLH 13 ppm |
| Stability | Stable. Substances to be avoided include strong oxidising agents, acids, peroxides and oxygen. Highly flammable; can form explosive mixtures with air. |
| InChIKey | YLQBMQCUIZJEEH-UHFFFAOYSA-N |
| LogP | 1.34 |
| CAS DataBase Reference | 110-00-9(CAS DataBase Reference) |
| IARC | 2B (Vol. 63) 1995 |
| NIST Chemistry Reference | Furan(110-00-9) |
| EPA Substance Registry System | Furan (110-00-9) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS02,GHS07,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H224-H302+H332-H315-H341-H350-H373-H412 | |||||||||
| Precautionary statements | P210-P233-P273-P304+P340+P312-P308+P313-P403+P233 | |||||||||
| Hazard Codes | T,F+ | |||||||||
| Risk Statements | 45-12-19-20/22-38-48/22-52/53-68 | |||||||||
| Safety Statements | 53-45-61- | |||||||||
| RIDADR | UN 2811 6.1/PG 2 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | OB3870000 | |||||||||
| F | 8-9-23 | |||||||||
| Autoignition Temperature | 390 °C | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 2932 19 00 | |||||||||
| HazardClass | 3 | |||||||||
| PackingGroup | I | |||||||||
| Toxicity | LC (in air) in rats: 30400 ppm (Henderson) | |||||||||
| IDLA | 13 ppm (35.1 mg/m3) | |||||||||
| NFPA 704 |
|
Furan price More Price(5)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 8.20594 | Furan (stabilised) for synthesis | 110-00-9 | 250ML | ₹4689.99 | 2022-06-14 | Buy |
| ALFA India | ALF-A13102-AP | Furan, 99%, stab. with ca 250ppm BHT | 110-00-9 | 500mL | ₹4213 | 2022-05-26 | Buy |
| ALFA India | ALF-A13102-AE | Furan, 99%, stab. with ca 250ppm BHT | 110-00-9 | 100mL | ₹2806 | 2022-05-26 | Buy |
| ottokemi | F 1560 | Furan 98% | 110-00-9 | 100mL | ₹3006 | 2022-05-26 | Buy |
| ottokemi | F 1560 | Furan 98% | 110-00-9 | 500mL | ₹8604 | 2022-05-26 | Buy |
Furan Chemical Properties,Uses,Production
Description
Furan occurs naturally in oils distilled from rosin containing pinewood. In addition, many natural foods contain the furan ring structure and substituted furans may be formed through cooking of simple carbohydrates. Furan is also found in tobacco smoke as well as wood smoke and gas emissions from gasoline and diesel engines. Furan has also been detected in industrial effluents and can be emitted to the air from petroleum refineries and coal-mining and gasification plants.
Chemical Properties
Furan is a cyclic dienic ether stabilized by benzene-like resonance. Because of its conjugated unsaturation and heterocyclic atom, furan will undergo many types of reactions. It is, therefore, of interest as a chemical intermediate for pharmaceuticals, insecticides and fine chemicals. The heterocyclic oxygen atom in a ring with conjugated unsaturation gives furan a combination of ether, aromatic and olefinic characteristics. This polyfunctionality permits it to undergo a variety of reactions. Compared to benzene, the furan ring has greater reactivity, and is more susceptible to cleavage, thus resembling the vinyl ethers. Like the vinyl ethers, the furan ring is cleaved by aqueous acids. This reaction is accompanied by resinification .
Uses
Furan is a five-membered heterocyclic aromatic ring. Furan is used as a building block for the preparation of many heterocyclic compounds.
Uses
In organic syntheses. Furan is occasionally used in the composing of artificial essential oils. It finds a little - rare - use in industrial fragrances for certain masking purposes, e. g. when the problem is: to mask very volatile, undesirable odors. It is used in special fragrances such as “smokey” odors for certain products, directly or indirectly connected with food products.
Definition
A heterocyclic liquid organic compound. Its five-membered ring contains four carbon atoms and one oxygen atom. The structure is characteristic of some monosaccharide sugars (furanoses).
Preparation
By decarboxylation of Furoic acid. Furan can also be prepared directly from Furfural.
General Description
Furan is a stable, colourless chemical liquid. It is highly flammable and forms explosive mixtures with air. It is incompatible with many other chemical substances, for example, strong oxidising agents, acids, peroxides, and oxygen. Furan is produced commercially by decarbonylation of furfural. It is used mainly in the production of tetrahydrofuran, thiophene, and pyrrole. It also occurs naturally in certain woods and during the combustion of coal and is found in engine exhausts, wood smoke, and tobacco smoke.
Air & Water Reactions
Highly flammable. When uninhibited, Furan forms explosive peroxides on exposure to air. Insoluble in water.
Reactivity Profile
Furan is sensitive to heat and may turn brown upon standing. Furan may be light sensitive. When uninhibited, Furan forms explosive peroxides on exposure to air. Furan may react with oxidizers, acids, peroxides and oxygen. Furan resinifies on evaporation or when in contact with mineral acids, but Furan is stable in alkalis. .
Hazard
Flammable, dangerous fire risk, flammable limits 2–24%, forms peroxides on exposure to air. Absorbed by skin. Possible carcinogen.
Health Hazard
The vapors are narcotic. Acute exposure to Furan by inhalation may involve both reversible and irreversible changes. Acute exposure by ingestion or skin absorption, as well as chronic exposure, are associated with high toxicity.
Fire Hazard
Very dangerous, upon exposure to heat or flame. Furan may form unstable peroxides on exposure to air. Contact with acids can initiate a violent, heat producing reaction. Avoid acids, oxidizing agents. Upon standing in air, Furan may form unstable peroxides.
Safety Profile
Confirmed carcinogen. Poison by inhalation and intraperitoneal routes. Moderately toxic by ingestion and skin contact. Experimental reproductive effects. A narcotic. Mutation data reported. The exposure concentration limit of 10 ppm together with its low boiling point requires that adequate ventilation be provided in areas where ths chemical is handled. Contact with liquid must be avoided since this chemical can be absorbed through the skin. Washing thoroughly with soap and water followed by prolonged rinsing should be done immedlatelp after accidental contact. A very dangerous fire hazard when exposed to heat or flame; can react with oxidzing materials. Unstabdized, it may form unstable peroxides on exposure to air and should always be tested before ddlation. Washing with an aqueous solution of ferrous sulfate slightly acidified with sodum bisulfate will remove these peroxides. Confirm by test. Contact with acids can initiate a violent exothermic reaction. Moderate explosion hazard when exposed to flame. Furan's low bohg point makes it easy to obtain explosive concentrations of the vapor in inadequately ventilated areas. To fight fire, use CO2, dr). chemical. When heated to decomposition it emits acrid smoke and irritating fumes. See also PEROXIDES.
Potential Exposure
Furan is used as a chemical intermedi ate in the production of herbicides and pharmaceuticals; for making tetrahydrofuran; in formation of lacquers; as a sol vent for resins in organic synthesis, especially for pyrrole, thiophene.
Carcinogenicity
Furan is reasonably anticipated to be a human carcinogen based on sufficient evidence of carcinogenicity from studies in experimental animals.
Environmental Fate
Furan may be released to the environment as a waste industrial product or from unintentional or accidental releases. If released to soil, it is expected to volatilize. If released to water, furan is not expected to adsorb to suspended particles and sediment and is likely to volatilize to ambient air. Sulfate-reducing bacteria can degrade furan. However, under nonsulfate-reducing conditions, biodegradation in soil and water is expected to be slow. In the air, furan will exist as a vapor and will be subject to degradation by reacting with hydroxyl radicals.
Shipping
UN2389 Furan, Hazard Class: 3; Labels: 3-Flammable liquid.
Purification Methods
Shake it with aqueous 5% KOH, dry it with CaSO4 or Na2SO4, then distil it under nitrogen, from KOH or sodium, immediately before use. A trace of hydroquinone could be added as an inhibitor of oxidation. [Beilstein 17 H 27, 17 I 16, 17 II 34, 17/1 V 291.]
Incompatibilities
May form explosive mixture with air. Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong acids, amines, strong bases, reducing agents. Unless stabilized with an inhibitor, air exposure forms unstable peroxides.
Waste Disposal
Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste containing this contaminant (≥100 kg/mo) must conform with EPA regulations govern ing storage, transportation, treatment, and waste disposal.
Furan Preparation Products And Raw materials
Raw materials
Preparation Products
1of5
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| Galaxy Laboratories Private Limited | +91-9850500858 +91-9850500858 | Maharashtra, India | 15 | 58 | Inquiry |
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| Otto Chemie Pvt. Ltd. | +91 9820041841 | Mumbai, India | 5873 | 58 | Inquiry |
| Alfa Aesar | 1 800 209 7001 | Maharashtra, India | 6913 | 58 | Inquiry |
| Central Drug House(P) Ltd. | 91-11-49404040 | New Delhi, India | 6160 | 58 | Inquiry |
| Ravi Refractories Corporation | 08048371520Ext 905 | Maharashtra, India | 1 | 58 | Inquiry |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | China | 21675 | 55 | Inquiry |
| SHANDONG ZHI SHANG CHEMICAL CO.LTD | +86 18953170293 | China | 2931 | 58 | Inquiry |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 | China | 49391 | 58 | Inquiry |
Related articles
- Electrophilic Reactions of Furan
- Furans are fairly soluble in organic solvents, including alcohol and ether, but slightly soluble in water. They are toxic and ....
- Jan 24,2022
- Uses of Furan
- Furan is a five-membered aromatic oxygen heterocycle, comprised of four conjugated carbon atoms and one oxygen atom in a cycli....
- Jan 24,2022
- Toxicity and hazards of Furan
- Furan can cause eye, skin, and mucus membrane irritation, burning sensation and, in severe cases, corrosion. If inhaled, furan....
- Oct 27,2021
Related Qustion
- Q:Furan in Foods
- A:Furan is a colorless, volatile liquid used in some chemical manufacturing industries. Furan has occasionally been reported to ....
- Sep 20,2019
110-00-9(Furan)Related Search:
1of4
chevron_right




