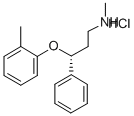
Atomoxetine hydrochloride
- CAS No.
- 82248-59-7
- Chemical Name:
- Atomoxetine hydrochloride
- Synonyms
- ATOMOXETINE;Strattera;ATOMOXETINE HCL;ATOMOXITINE;Atomoxetine HCI;LY 139603;LY 139603 HCl;Atazanavir HCl;Atomoxitine HCL;(R)-Atomoxetine HCl
- CBNumber:
- CB5757860
- Molecular Formula:
- C17H22ClNO
- Molecular Weight:
- 291.82
- MOL File:
- 82248-59-7.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/7/31 13:51:23
| Melting point | 167-169°C |
|---|---|
| alpha | D25 -38.01°; 36525 -177.26° (c = 1 in methanol); D23 -41.37° (c = 1.02 in methanol); D25 -40.3° (c = 0.94 in ethanol) |
| Flash point | 9℃ |
| storage temp. | 2-8°C |
| solubility | soluble in Methanol |
| form | solid |
| pka | 10.13(at 25℃) |
| color | White to Almost white |
| Water Solubility | Soluble to 50 mM in water with gentle warming |
| Merck | 14,863 |
| BCS Class | 1? |
| InChI | InChI=1/C17H21NO.ClH/c1-14-8-6-7-11-16(14)19-17(12-13-18-2)15-9-4-3-5-10-15;/h3-11,17-18H,12-13H2,1-2H3;1H/t17-;/s3 |
| InChIKey | LUCXVPAZUDVVBT-UNTBIKODSA-N |
| SMILES | O([C@H](CCNC)C1C=CC=CC=1)C1=CC=CC=C1C.Cl |&1:1,r| |
| CAS DataBase Reference | 82248-59-7(CAS DataBase Reference) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS02,GHS06,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H225-H301+H311+H331-H370 | |||||||||
| Precautionary statements | P210-P260-P280-P301+P310-P311 | |||||||||
| Hazard Codes | F,T | |||||||||
| Risk Statements | 11-23/24/25-39/23/24/25 | |||||||||
| Safety Statements | 22-24/25-45-36/37-16-7 | |||||||||
| RIDADR | UN1230 - class 3 - PG 2 - Methanol, solution | |||||||||
| WGK Germany | 3 | |||||||||
| HS Code | 29221990 | |||||||||
| NFPA 704 |
|
Atomoxetine hydrochloride price More Price(6)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | T7947 | (R)-Tomoxetine hydrochloride solid | 82248-59-7 | 25MG | ₹58898.83 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | T7947 | (R)-Tomoxetine hydrochloride solid | 82248-59-7 | 100MG | ₹148832.93 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | PHR1679 | Atomoxetine Hydrochloride Pharmaceutical Secondary Standard; Certified Reference Material | 82248-59-7 | 500MG | ₹16205.03 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | A-095 | Atomoxetine hydrochloride solution 1.0?mg/mL in methanol (as free base), ampule of 1?mL, certified reference material, Cerilliant? | 82248-59-7 | 1ML | ₹16837.8 | 2022-06-14 | Buy |
| TCI Chemicals (India) | A2357 | Atomoxetine Hydrochloride | 82248-59-7 | 100MG | ₹5500 | 2022-05-26 | Buy |
Atomoxetine hydrochloride Chemical Properties,Uses,Production
Description
Atomoxetine is the first non-stimulant marketed for the treatment of attention deficit hyperactivity disorder (ADHD). It is the R-stereoisomer of the racemate tomoxetine and is a selective and potent norepinephrine uptake inhibitor (Ki=0.7–1.9 nM) that is devoid of binding to monoamine receptor. It also has little effect on dopamine and serotonin reuptake or acetylcholine, H1 histamine, alpha1 or alpha1-adrenergic or dopamine receptors. It is prepared from racemic 1-phenylbut-3-en-1-ol via a selective enzymatic acylation leaving the desired S-stereoisomer as the alcohol. This alcohol is converted via a Mitsunobu reaction with ortho-cresol to the corresponding ether with isomeric R-configuration. Ozonolysis and reduction steps provided the terminal alcohol that is mesylated and displaced with methylamine. Its selectivity for norepinephrine relative to dopamine inhibition was demonstrated in vivo preclinically. In a two-lever (two condition) discriminative stimulus effect study in squirrel monkeys, tomoxetine and other norepinephrine uptake inhibitors substituted for cocaine under low-dose training conditions, whereas dopamine uptake inhibitors substituted for cocaine in both low and high-dose conditions. In clinical ADHD studies in adolescents, it was significantly different from placebo in 1.2 and 1.8 mpk/day dosing. In the clinical study in adults using the CAARS scale a 95 mg/day dose provided greater than 30% improvement in total scores. Atomoxetine is about 63% orally bioavailable, is highly protein bound (98%, primarily to albumin) and has a half-life of about 5.2 h. It is metabolized by CYP2D6 resulting in differential clearance for poor metabolizers (halflife of 19 h with a 10 times higher AUC) relative to extensive metabolizers. The total daily dose for children, adolescents and adults is a maximum of 100 mg/day. Common side effects in children and adults include nausea, decreased appetite, and dizziness. Adults may also have insomnia.
Chemical Properties
White Solid
Uses
These Secondary Standards are qualified as Certified Reference Materials. These are suitable for use in several analytical applications including but not limited to pharma release testing, pharma method development for qualitative and quantitative analyses, food and beverage quality control testing, and other calibration requirements.
Atomoxetine hydrochloride may be used as a pharmaceutical reference standard for the determination of atomoxetine hydrochloride in pharmaceutical formulations by spectrophotometric method.
Definition
ChEBI: The hydrochloride salt of atomoxetine.
General Description
Atomoxetine hydrochloride is a nonstimulant used in the treatment of Attention-Deficit/Hyperactivity Disorder (ADHD) in children and adults.
Biological Activity
Potent and selective noradrenalin re-uptake inhibitor (K i values are 5, 77 and 1451 nM for inhibition of radioligand binding to human NET, SERT and DAT respectively). Displays minimal affinity for a range of other neurotransmitter receptors and transporters (K i > 1 μ M). Antidepressant.
Atomoxetine hydrochloride Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Cadila Pharmaceuticals Ltd | +91-7069076657 +91-7069076657 | Ahmedabad, India | 54 | 58 | Inquiry |
| Dr. Reddy's Laboratories Ltd | +91-4049002900 +91-4049002900 | Hyderabad, India | 165 | 58 | Inquiry |
| Global Pharma | +91-9819994077 +91-9819994077 | Maharashtra, India | 34 | 58 | Inquiry |
| KARPSCHEM LABORATORIES | +91-7249203006 +91-7249203006 | Maharashtra, India | 786 | 58 | Inquiry |
| Aurobindo Pharma Limited | +914066725000 | Telangana, India | 112 | 58 | Inquiry |
| R L Fine Chem Pvt. Ltd | +91-8042488999 +91-8042488999 | Karnataka, India | 74 | 58 | Inquiry |
| Hetero Drugs Limited | +91-4023704923 +91-4023704923 | Telangana, India | 296 | 58 | Inquiry |
| Unichem Laboratories Ltd | +91-2266888333 +91-2266888333 | Maharashtra, India | 62 | 58 | Inquiry |
| Vasoya Industries Pvt Ltd | +91-9898083583 +91-9898083583 | Gujarat, India | 42 | 58 | Inquiry |
| ZCL Chemicals Ltd | +91-2261539999 +91-2261539999 | Maharashtra, India | 53 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| Cadila Pharmaceuticals Ltd | 58 |
| Dr. Reddy's Laboratories Ltd | 58 |
| Global Pharma | 58 |
| KARPSCHEM LABORATORIES | 58 |
| Aurobindo Pharma Limited | 58 |
| R L Fine Chem Pvt. Ltd | 58 |
| Hetero Drugs Limited | 58 |
| Unichem Laboratories Ltd | 58 |
| Vasoya Industries Pvt Ltd | 58 |
| ZCL Chemicals Ltd | 58 |
82248-59-7(Atomoxetine hydrochloride)Related Search:
1of4
chevron_right




