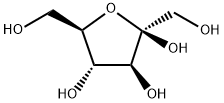
D(-)-Fructose
- CAS No.
- 57-48-7
- Chemical Name:
- D(-)-Fructose
- Synonyms
- FRUCTOSE;D-FRUCTOSE;Fluctose;D-(-)-Fructose 〔Levulose〕;Fructose Standard, 18000ppm;Krystar;d-fructos;LAEVULOSE;Fructose CRS;D-(-)-Fructose ,99% [Natural]
- CBNumber:
- CB6139083
- Molecular Formula:
- C6H12O6
- Molecular Weight:
- 180.16
- MOL File:
- 57-48-7.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/6/17 17:42:23
| Melting point | 119-122 °C (dec.)(lit.) |
|---|---|
| alpha | -92.25 º (c=10,H2O,on dry sub.) |
| Boiling point | 232.96°C (rough estimate) |
| Density | 1.59 |
| refractive index | -92 ° (C=4, H2O) |
| storage temp. | room temp |
| solubility | H2O: 1 M at 20 °C, clear, colorless |
| pka | pKa (18°): 12.06 |
| form | Crystals or Crystalline Powder |
| color | White |
| PH | 5.0-7.0 (25℃, 0.1M in H2O) |
| Odor | at 100.00 %. odorless |
| Odor Type | odorless |
| optical activity | [α]20/D 93.5 to 91.0°, c = 10% in H2O |
| Water Solubility | 3750 g/L (20 ºC) |
| λmax |
λ: 260 nm Amax: 0.04 λ: 280 nm Amax: 0.04 |
| Merck | 14,4273 |
| BRN | 1239004 |
| Stability | Stable. Incompatible with strong oxidizing agents. |
| InChIKey | LKDRXBCSQODPBY-GWVKGMJFSA-N |
| LogP | -1.029 (est) |
| CAS DataBase Reference | 57-48-7(CAS DataBase Reference) |
| NIST Chemistry Reference | «beta»-D-Fructose(57-48-7) |
| EPA Substance Registry System | D-Fructose (57-48-7) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H315-H319-H335 | |||||||||
| Precautionary statements | P261-P271-P280 | |||||||||
| Hazard Codes | C | |||||||||
| Risk Statements | 34 | |||||||||
| Safety Statements | 24/25-45-36/37/39-27-26 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | LS7120000 | |||||||||
| F | 3 | |||||||||
| Autoignition Temperature | 360 °C | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 17025000 | |||||||||
| NFPA 704 |
|
D(-)-Fructose price More Price(38)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | F9048 | D-(−)-Fructose meets USP testing specifications | 57-48-7 | 100G | ₹5413.1 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | F9048 | D-(−)-Fructose meets USP testing specifications | 57-48-7 | 500G | ₹5758.9 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | PHR1002 | Fructose Pharmaceutical Secondary Standard; Certified Reference Material | 57-48-7 | 1G | ₹10267.6 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | F9048 | D-(−)-Fructose meets USP testing specifications | 57-48-7 | 1KG | ₹9443 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | F3510 | D-(−)-Fructose BioReagent, suitable for cell culture, suitable for insect cell culture | 57-48-7 | 100G | ₹2899.4 | 2022-06-14 | Buy |
D(-)-Fructose Chemical Properties,Uses,Production
Chemical Properties
White Cyrstalline Solid
History
Despite this ubiquity, fructose remained a noncommercial product until the 1980s because of the expense involved in its isolation and the care required for its handling. The development of technologies for preparing fructose from glucose in the isomerized mixture led to a greater availability of pure, crystalline fructose in the 1970s. However, the price for pure fructose was high enough in 1981 that the product was not competitive with sucrose and corn syrups as a commercial sweetener. With the entry of corn wet-milling companies into the crystalline fructose market in the late 1980s, raw material economies and enlarged manufacturing scale led to a nearly 10-fold production increase within a five-year period, making fructose prices competitive with other sweeteners for specific applications.
Uses
D-Fructose occurs in a large number of fruits, honey, and as the sole sugar in bull and human semen
Definition
A sugar found in fruit juices, honey, and cane sugar. It is a ketohexose, existing in a pyranose form when free. In combination (e.g. in sucrose) it exists in the furanose form.
Production Methods
Fructose, a monosaccharide sugar, occurs naturally in honey and a large number of fruits. It may be prepared from inulin, dextrose, or sucrose by a number of methods. Commercially, fructose is mainly manufactured by crystallization from high-fructose syrup derived from hydrolyzed and isomerized cereal starch or cane and beet sugar.
General Description
Fructose is a monosaccharide. It is present in fruits and vegetables. Fructose is the major carbohydrate in the diet. It binds with glucose to form sucrose. Excessive intake of fructose is associated with obesity, type 2 diabetes and cardiovascular disease.
Safety
Although it is absorbed more slowly than dextrose from the
gastrointestinal tract, fructose is metabolized more rapidly. Metabolism
of fructose occurs mainly in the liver, where it is converted
partially to dextrose and the metabolites lactic acid and pyruvic
acid. Entry into the liver and subsequent phosphorylation is insulinindependent.
Further metabolism occurs by way of a variety of
metabolic pathways. In healthy and well regulated diabetics,
glycogenesis (glucose stored as glycogen) predominates.
Excessive oral fructose consumption (>75 g daily) in the absence
of dietary dextrose in any form (e.g. sucrose, starch, dextrin, etc.)
may cause malabsorption in susceptible individuals, which may
result in flatulence, abdominal pain, and diarrhea. Except in
patients with hereditary fructose intolerance, there is no
evidence to indicate that oral fructose intake at current levels is a
risk factor in any particular disease, other than dental caries.
storage
Fructose is hygroscopic and absorbs significant amounts of
moisture at relative humidities greater than 60%. Goods stored in
the original sealed packaging at temperatures below 25°C and a
relative humidity of less than 60% can be expected to retain stability
for at least 12 months.
Aqueous solutions are most stable at pH 3–4 and temperatures
of 4–70°C; they may be sterilized by autoclaving.
Purification Methods
Dissolve D(-)-fructose in an equal weight of water (charcoal, previously washed with water to remove any soluble material), filter and evaporate under reduced pressure at 45-50o to give a syrup containing 90% of fructose. After cooling to 40o, the syrup is seeded and kept at this temperature for 20-30hours with occasional stirring. The crystals are removed by centrifugation, washed with a small quantity of water and dried to constant weight under a vacuum over conc H2SO4. For higher purity, this material is recrystallised from 50% aqueous ethanol [Tsuzuki et al. J Am Chem Soc 72 1071 1950]. [Beilstein 31 H 321, 1 IV 4401.]
Incompatibilities
Incompatible with strong acids or alkalis, forming a brown coloration. In the aldehyde form, fructose can react with amines, amino acids, peptides, and proteins. Fructose may cause browning of tablets containing amines.
Regulatory Status
Included in the FDA Inactive Ingredients Database (oral solutions, syrup, and suspensions; rectal preparations; intravenous infusions). Included in the Canadian List of Acceptable Non-medicinal Ingredients.
D(-)-Fructose Preparation Products And Raw materials
Raw materials
1of4
chevron_rightPreparation Products
1of2
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| UNILOSA INTERNATINAL PRIVATE LIMITED | +91-9999069917 +91-9999069917 | New Delhi, India | 152 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| Merck Ltd | +91-2262109800 +91-2262109000 | Maharashtra, India | 272 | 58 | Inquiry |
| SOLFYN INTERNATIONAL LLP | +91-9321772608 +91-9321772608 | Mumbai, India | 105 | 58 | Inquiry |
| S D Fine Chem Limited | +91-9323715856 +91-9323715856 | Maharashtra, India | 368 | 58 | Inquiry |
| Titan Biotech Limited | 09711169006 | Delhi, India | 196 | 58 | Inquiry |
| Advent Chembio Pvt., Ltd. | 91-22-27690836 | Maharashtra, India | 625 | 58 | Inquiry |
| Anand Agencies | 91-20-24454597 | Maharashtra, India | 2337 | 58 | Inquiry |
| 5 E Scientific | 91-22-28016026 | Maharashtra, India | 137 | 58 | Inquiry |
| Opulent Pharma | 08068441147Ext 981 | Gujarat, India | 810 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| UNILOSA INTERNATINAL PRIVATE LIMITED | 58 |
| JSK Chemicals | 58 |
| Merck Ltd | 58 |
| SOLFYN INTERNATIONAL LLP | 58 |
| S D Fine Chem Limited | 58 |
| Titan Biotech Limited | 58 |
| Advent Chembio Pvt., Ltd. | 58 |
| Anand Agencies | 58 |
| 5 E Scientific | 58 |
| Opulent Pharma | 58 |





