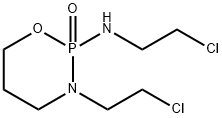
Ifosfamide
- CAS No.
- 3778-73-2
- Chemical Name:
- Ifosfamide
- Synonyms
- IF;ISOPHOSPHAMIDE;Ifosfamide sterile;3-(2-chloroethyl)-2-[(2-chloroethyl)amino]tetrahydro-2h-1,3,2-oxazaphosphorine 2-oxide;Ifex;mitoxana;z4942;cyfos;A-4942;mjf9325
- CBNumber:
- CB7489350
- Molecular Formula:
- C7H15Cl2N2O2P
- Molecular Weight:
- 261.09
- MOL File:
- 3778-73-2.mol
- MSDS File:
- SDS
- Modify Date:
- 2025/2/27 17:19:27
| Melting point | 48°C |
|---|---|
| Boiling point | 336.1±52.0 °C(Predicted) |
| Density | 1.33±0.1 g/cm3(Predicted) |
| storage temp. | Inert atmosphere,2-8°C |
| solubility |
DMF:50.0(Max Conc. mg/mL);191.51(Max Conc. mM) DMSO:44.0(Max Conc. mg/mL);168.53(Max Conc. mM) Ethanol:51.0(Max Conc. mg/mL);195.34(Max Conc. mM) PBS (pH 7.2):10.0(Max Conc. mg/mL);38.3(Max Conc. mM) Water:52.0(Max Conc. mg/mL);199.17(Max Conc. mM) |
| pka | 1.44±0.20(Predicted) |
| form | Solid |
| color | White to off-white |
| CAS DataBase Reference | 3778-73-2(CAS DataBase Reference) |
| IARC | 3 (Vol. 26, Sup 7) 1987 |
| EPA Substance Registry System | Isophosphamide (3778-73-2) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS06,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H301-H319-H340-H350-H360 | |||||||||
| Precautionary statements | P201-P301+P310+P330-P305+P351+P338 | |||||||||
| Hazard Codes | T | |||||||||
| Risk Statements | 25-36 | |||||||||
| Safety Statements | 26-45 | |||||||||
| RIDADR | 3249 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | RP6050000 | |||||||||
| HazardClass | 6.1(b) | |||||||||
| PackingGroup | III | |||||||||
| HS Code | 29349990 | |||||||||
| Hazardous Substances Data | 3778-73-2(Hazardous Substances Data) | |||||||||
| Toxicity | LD50 in rats (mg/kg): 160 i.p. (Arnold, 1973); also reported as 150 i.p. (Brock) | |||||||||
| NFPA 704 |
|
Ifosfamide price More Price(3)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | I4909 | Ifosfamide ≥98% | 3778-73-2 | 1G | ₹17493.2 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | PHR3187 | Ifosfamide pharmaceutical secondary standard, certified reference material | 3778-73-2 | 1G | ₹19625.73 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | I4909 | Ifosfamide ≥98% | 3778-73-2 | 5G | ₹54384.8 | 2022-06-14 | Buy |
Ifosfamide Chemical Properties,Uses,Production
Chemical Properties
Crystalline Solid
Uses
A cytostatic agent, related structurally to cyclophosphamide
Indications
Ifosfamide (Ifex) is an analogue of cyclophosphamide
that requires metabolic activation to form 4-hydroxyifosfamide.
In general, the metabolism, serum half-life,
and excretion of ifosfamide are similar to those of cyclophosphamide.
Ifosfamide is active against a broad spectrum of tumors,
including germ cell cancers of the testis, lymphomas,
sarcomas, and carcinomas of the lung, breast,
and ovary. It is thought to be more active than cyclophosphamide
in germ cell cancers and sarcomas.
Ifosfamide is less myelosuppressive than cyclophosphamide
but is more toxic to the bladder. It also may
produce alopecia, nausea, vomiting, infertility, and second
tumors, particularly acute leukemias. Neurological
symptoms including confusion, somnolence, and hallucinations
have also been reported. It is recommended
that ifosfamide be coadministered with the thiol compound
mesna (Mesnex) to avoid hemorrhagic cystitis.
Definition
ChEBI: The simplest member of the class of ifosfamides that is 1,3,2-oxazaphosphinan-2-amine 2-oxide substituted by 2-chloroethyl groups on both the nitrogen atoms respectively. It is a nitrogen mustard alkylating agent used in the treatment of advanced breast c ncer.
General Description
Ifosfamide is available in 1- and 3-g vials for IV administrationas Food and Drug Administration (FDA)-approvedthird-line therapy in the treatment of testicular cancer. It has also been utilized (although not FDA approved) in the treatmentof a wide variety of cancers including Hodgkin’s andnon-Hodgkin’s lymphoma, soft tissue sarcoma, germ celltumors, small cell lung cancer, non–small cell lung cancer(NSCLC), cancers of the head and neck, bladder cancer, cervicalcancer, and Ewing sarcoma. Coadministration ofmesna is recommended. The mechanisms of resistance areidentical to those seen with cyclophosphamide. The drug iswidely distributed with a low extent of protein binding(20%). Metabolism primarily by CYP3A4/5 and CYP2B6 isrequired for activation of the compound. Theagent is administered as the racemic mixture as a result ofthe presence of the chiral phosphorus atom, and differentialmetabolism of the R- and S-isomers has been observed. Incontrast to cyclophosphamide, there is a greater amount ofdeactivation of the agent by N-dechloroethylation and subsequentlymore chloroacetaldehyde is produced, which mayresult in a greater amount of neurotoxicity and nephrotoxicitythan seen with cyclophosphamide. The N-dechloroethylatedmetabolites are the predominate species seen in theplasma. The parent and metabolites are eliminated inthe urine with an elimination half-life of 3 to 10 hours. Themajor components found in the urine are the dechlorethylatedmetabolites. Dose-limiting toxicities include myelosuppressionand bladder toxicity. Other adverse effectsinclude nausea, alopecia, amenorrhea, inappropriate secretionof antidiuretic hormone, as well as the production ofsecondary cancers. Neurotoxicity, which is associated withthe production of chloroacetaldehyde presents as confusion,seizure, weakness, and hallucination, and coma may occur.
Clinical Use
Ifosamide currently is used as “third-line” therapy against testicular cancer, although it also has shown activity in a number of solid tumors and hematologic cancers.
Side effects
Patients on ifosfamide (but not cyclophosphamide) commonly exhibit central nervous system (CNS) toxicity. In severe forms, CNS depression can progress to coma and death.
Ifosfamide Preparation Products And Raw materials
Raw materials
1of2
chevron_rightPreparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| KROMOZOME | +91-91-9650900955 +91-09650900955 | Maharashtra, India | 71 | 58 | Inquiry |
| SAMSON ORGANICS | +91-8341971897 +91-8341971897 | Hyderabad, India | 15 | 58 | Inquiry |
| Aarti Industries Limited (AIL) | +91-84596144931 +91-9920899935 | Maharashtra, India | 235 | 58 | Inquiry |
| Basil Drugs AND Pharmaceuticals Pvt Ltd | +91-2249700250 +91-9619320820 | Mumbai, India | 108 | 58 | Inquiry |
| Ralington Pharma | +91-7948911722 +91-9687771722 | Gujarat, India | 1350 | 58 | Inquiry |
| Sakar Healthcare | +91-8976292690 +91-9967572302 | Gujarat, India | 47 | 58 | Inquiry |
| Aspen Biopharma Labs Pvt Ltd | +91-9248058660 +91-9248058662 | Telangana, India | 234 | 58 | Inquiry |
| Kekule Life Sciences Limited | +91-9963474090 +91-9963474090 | AndhraPradesh, India | 19 | 58 | Inquiry |
| Vyshno Bio Sciences | 91-7095439993 | Hyderabad, India | 239 | 58 | Inquiry |
| AARTI INDUSTRIES LTD | +91 22 6797 6666 | New Delhi, India | 32 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| KROMOZOME | 58 |
| SAMSON ORGANICS | 58 |
| Aarti Industries Limited (AIL) | 58 |
| Basil Drugs AND Pharmaceuticals Pvt Ltd | 58 |
| Ralington Pharma | 58 |
| Sakar Healthcare | 58 |
| Aspen Biopharma Labs Pvt Ltd | 58 |
| Kekule Life Sciences Limited | 58 |
| Vyshno Bio Sciences | 58 |
| AARTI INDUSTRIES LTD | 58 |
3778-73-2(Ifosfamide)Related Search:
1of4
chevron_right




