Sorafenib tosylate
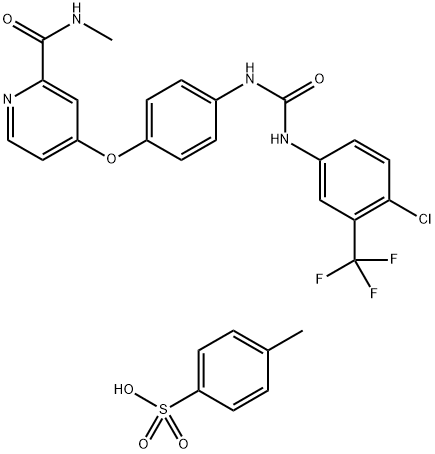
- CAS No.
- 475207-59-1
- Chemical Name:
- Sorafenib tosylate
- Synonyms
- CL050;Sorafenib &Sorafeni tosylate;Sorafinib mesylate;SORAFENIB TOSYLATE;Sorafenib (Nexavar);Sorafenib TsOH salt;Sorafenib 475207-59-1;Sorafenib tosylate API;BAY 43-9006;BAY43-9006
- CBNumber:
- CB92131200
- Molecular Formula:
- C21H16ClF3N4O3.C7H8O3S
- Molecular Weight:
- 637.03
- MOL File:
- 475207-59-1.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/4/12 23:00:59
| Melting point | 229-232°C |
|---|---|
| storage temp. | 2-8°C(protect from light) |
| solubility | Soluble in DMSO (up to 200 mg/ml) or in Ethanol (up to 3 mg/ml). |
| form | solid |
| color | Off-white |
| Stability | Stable for 2 years from date of purchase as supplied. Solutions in DMSO or ethanol may be stored at -20° for up to 1 month. |
| InChIKey | IVDHYUQIDRJSTI-UHFFFAOYSA-N |
| SMILES | S(C1C=CC(C)=CC=1)(O)(=O)=O.O(C1C=CC(NC(=O)NC2C=CC(Cl)=C(C(F)(F)F)C=2)=CC=1)C1=CC=NC(C(=O)NC)=C1 |
| CAS DataBase Reference | 475207-59-1(CAS DataBase Reference) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS08,GHS09 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H360FD-H362-H372-H410 | |||||||||
| Precautionary statements | P201-P202-P260-P263-P273-P308+P313 | |||||||||
| Risk Statements | 36/37/38 | |||||||||
| Safety Statements | 28-26 | |||||||||
| HS Code | 29159000 | |||||||||
| NFPA 704 |
|
Sorafenib tosylate Chemical Properties,Uses,Production
Uses
Sorafenib Tosylate (Bay 43-9006, Nexavar) is a small molecular inhibitor of VEGFR, PDGFR, c-Raf and B-Raf with IC50s of 18 nM, 10 nM, 3 nM and 15 nM, respectively.
brand name
Nexavar (Bayer).
General Description
Sorafenib is available in 200-mg tablets for oral administrationand is used in the treatment of RCC and colon cancer.The agent is classified as a multikinase inhibitor because ofits action on numerous kinase enzymes including thePDGF-R, VEGF-R, Kit, and Raf. Sorafenib is 39% to 48% bioavailable and CYP3A4-mediated metabolism giveseight identified metabolites including the N-oxide, which isequally active with the parent. However, the majority of thedrug in plasma is present as the parent compound.Sorafenib is highly protein bound (99.5%). The drug iseliminated primarily in the feces (77%) with 19% appearingin the urine as glucuronides (UGT1A1) and has a eliminationhalf-life of 24 to 48 hours. The most commonly seentoxicity is skin rash that normally occurs within the first6 weeks of therapy. Other adverse effects include hypertension,fatigue, increased wound healing time, and increasedrisk of bleeding.
Clinical Use
Sorafenib is used to treat late-stage kidney cancer (advanced renal cell carcinoma), liver cancer (hepatocellular carcinoma) that cannot be treated by surgery, and differentiated thyroid cancer that has come back or spread to other parts of your body. Sorafenib is an antineoplastic (cancer) agent. It interferes with the growth of cancer cells, which are eventually destroyed by the body.
Side effects
The side effects of Sorafenib tosylate include Bleeding gums, blistering, peeling, redness, or swelling of the palms of the hands or bottoms of the feet, bloating of the abdomen or stomach, blood in the urine or stools, clay-colored stools, coughing up blood, difficulty with breathing or swallowing, increased menstrual flow or vaginal bleeding.
Sorafenib tosylate Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Cipla Ltd | +912224826000 | Maharashtra, India | 133 | 58 | Inquiry |
| Alembic Pharmaceuticals Limited | +91-2652280880 +91-2652280550 | Gujarat, India | 115 | 58 | Inquiry |
| Vannsh Life Sciences Pvt Ltd | +91-4023555246 +91-9642555246 | Hyderabad, India | 15 | 58 | Inquiry |
| Shilpa Medicare Limited (SML) | +91-8532238704 +91-9320649838 | Karnataka, India | 61 | 58 | Inquiry |
| Raising Sun Pharma | +91-9399941155 +91-9399941155 | Telangana, India | 127 | 58 | Inquiry |
| Ralington Pharma | +91-7948911722 +91-9687771722 | Gujarat, India | 1350 | 58 | Inquiry |
| Brawn Laboratories Limited | +91-01129815331 +91-01129815331 | Haryana, India | 20 | 58 | Inquiry |
| Beta Drugs | +91-9316590666 +91-7015991921 | Haryana, India | 34 | 58 | Inquiry |
| Mac Chem Products (India) Pvt Ltd (Naprod Group) | +91-9727753234 +91-7770016606 | Maharashtra, India | 37 | 58 | Inquiry |
| Vishrudh laboratories pvt ltd | +91-9666132889 +91-8097369002 | Telangana, India | 117 | 58 | Inquiry |
Related articles
- Sorafenib Tosylate: Biological Activities and Future Developments
- Sorafenib tosylate, effective in hepatocellular carcinoma and renal cell carcinom, aims for enhanced efficacy through combinat....
- Mar 19,2024
- Sorafenib tosylate: mechanism of action, clinical applications and safety
- Sorafenib tosylate inhibits tumor angiogenesis, targeting cell proliferation and angiogenesis pathways. It shows promise as a ....
- Oct 9,2023
- About Sorafenib tosylate
- Sorafenib is used to treat kidney, liver, and thyroid cancer. It is a chemotherapy drug that works by slowing or stopping the ....
- Aug 14,2019
Related Qustion
- Q:When was sorafenib tosylate approved by the FDA?
- A:On December 20, 2005, the Food and Drug Administration (FDA) granted regular marketing approval for sorafenib tosylate 200-mg ....
- Apr 2,2024





