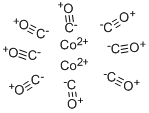
Cobalt carbonyl
- CAS No.
- 10210-68-1
- Chemical Name:
- Cobalt carbonyl
- Synonyms
- Co2(CO)8;DICOBALT OCTACARBONYL;DicobaL;t OctacarbonyL;cobaltcarbonyle;COBALT CARBONYL;dicobaltcarbonyl;cobaltoctacarbonyl;carbonyl,tetra-cobal;OCTACARBONYLDICOBALT
- CBNumber:
- CB9432426
- Molecular Formula:
- C8Co2O8+4
- Molecular Weight:
- 341.95
- MOL File:
- 10210-68-1.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/8/6 19:19:17
| Melting point | 51-52 °C |
|---|---|
| Boiling point | 52°C |
| Density | 1.81 |
| vapor pressure | 9.333-200Pa at 15-25℃ |
| Flash point | -13 °C |
| storage temp. | 2-8°C |
| solubility | Dichloromethane (Slightly), Methanol (Slightly) |
| form | crystal |
| Specific Gravity | 1.73 |
| color | dark orange |
| Water Solubility | Insoluble in water. Soluble in alcohol, ether and carbon disulfideSoluble in ether, naphtha and carbon disulfide. Slightly soluble in alcohol. Insoluble in water. |
| Sensitive | Air Sensitive |
| Hydrolytic Sensitivity | 7: reacts slowly with moisture/water |
| Merck | 13,3112 |
| Exposure limits |
TLV-TWA: 0.1 mg/m3 as Co (ACGIH) PEL-TWA: 0.1 mg/m3 as Co (OSHA). |
| Stability | Air sensitive |
| CAS DataBase Reference | 10210-68-1(CAS DataBase Reference) |
| EPA Substance Registry System | Cobalt carbonyl (10210-68-1) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS02,GHS06,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H251-H302-H315-H317-H330-H351-H361f-H373-H412 | |||||||||
| Precautionary statements | P201-P235-P273-P280-P301+P312+P330-P304+P340+P310 | |||||||||
| Hazard Codes | F,Xn,T+ | |||||||||
| Risk Statements | 11-22-40-43-48/20-52/53-62-26 | |||||||||
| Safety Statements | 36/37-61-45-28 | |||||||||
| RIDADR | UN 3190 4.2/PG 2 | |||||||||
| OEB | D | |||||||||
| OEL | TWA: 0.1 mg/m3 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | GG0300000 | |||||||||
| F | 23-25 | |||||||||
| TSCA | No | |||||||||
| HazardClass | 4.1 | |||||||||
| PackingGroup | II | |||||||||
| HS Code | 29319090 | |||||||||
| Toxicity | LD50 in mice, rats (mg/kg): 377.7, 753.8 by gavage (Spiridonova, Shabalina) | |||||||||
| NFPA 704 |
|
Cobalt carbonyl price More Price(7)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 60811 | Cobalt carbonyl moistened with hexane (hexane 1-10%), ≥90% (Co) | 10210-68-1 | 5G | ₹3734.63 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 60811 | Cobalt carbonyl moistened with hexane (hexane 1-10%), ≥90% (Co) | 10210-68-1 | 25G | ₹16389.05 | 2022-06-14 | Buy |
| ALFA India | ALF-013060-88 | Octacarbonyldicobalt, stab. with 1-5% hexane | 10210-68-1 | 2.5g | ₹3011 | 2022-05-26 | Buy |
| ALFA India | ALF-013060-22 | Octacarbonyldicobalt, stab. with 1-5% hexane | 10210-68-1 | 100g | ₹117464 | 2022-05-26 | Buy |
| ALFA India | ALF-013060-14 | Octacarbonyldicobalt, stab. with 1-5% hexane | 10210-68-1 | 25g | ₹30997 | 2022-05-26 | Buy |
Cobalt carbonyl Chemical Properties,Uses,Production
Chemical Properties
Cobalt carbonyl is a pyrophoric (spontaneously flammable in air), red-orange (when pure) to dark-brown crystalline solid.
Physical properties
Orange crystals; density 1.78 g/cm3; melts at 5l°C; decomposes above this temperature; insoluble in water; soluble in most organic solvents including alcohol, ether, carbon disulfide.
Uses
- Cobalt carbonyl [Co2(CO)8] is commonly used as a catalyst in the hydroformylation (oxo reaction) of alkenes.
- Along with pyridine, it can be used as a catalyst in the carboxylation of alkenes into corresponding acids and esters.
- It is employed as a key precursor in the preparation of cobalt platinum (CoPt3), cobalt sulfide (Co3S4) and cobalt selenide (CoSe2) nanocrystals.
- It is also used as a reagent in Pauson-Khand cyclizations and Nicholas reaction.
Preparation
Cobalt octacarbonyl is prepared by the reaction of finely divided cobalt with carbon monoxide under pressure:
2Co + 8CO → Co2(CO)8
The compound may be prepared in a similar way from cobalt(II) iodide. Also, it may be prepared by thermal decomposition of cobalt carbonyl hydride:
2HCo(CO)4 → Co2(CO)8 + H2
Hazard
Toxic by ingestion and inhalation.
Health Hazard
Dicobalt octacarbonyl exhibits moderate toxicityby inhalation route and somewhatlower toxicity by intraperitoneal and oralroutes. However, it is much less toxicthan nickel tetracarbonyl or iron pentacarbonyl.A 2-hour LC50 value in mice isreported as 27 mg/m3 (Lewis 1996). Anoral LD50 value in rats is within the rangeof 750–800 mg/kg. It decomposes, evolvingtoxic carbon monoxide.
Safety Profile
Poison by inhalation and intraperitoneal routes. Questionable carcinogen. Decomposes in air to form a product that ignites spontaneously in air. "hen heated to decomposition it emits acrid smoke and fumes. See also CARBONYLS and COBALT COMPOUNDS.
Potential Exposure
This material is used as a catalyst for a number of reactions. It is also used in antiknock gasoline and for high-purity cobalt salts.
Shipping
UN3124 Toxic solids, self-heating, n.o.s., Hazard Class: 6.1; 6.1-Poisonous materials, 4.2-Spontaneously combustible material. Technical Name Required. UN3190 Self-heating solid, inorganic, Hazard Class: 4.2; Labels: 4.2-Spontaneously combustible material, Technical Name Required. UN1325 Flammable solid, organic, n.o.s. Hazard Class: 4.1; Labels: 4.1-Flammable solid
Purification Methods
It forms orange-brown crystals on recrystallisation from n-hexane under a carbon monoxide atmosphere [Ojima et al. J Am Chem Soc 109 7714 1987; see also Hileman in Preparative Inorganic Reactions, Ed. Jolly, Vol 1 p 101 1987].
Incompatibilities
Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, epoxides. Decomposes on exposure to air or heat (@ ~52°C) producing toxic fumes of cobalt and oxides of carbon
Cobalt carbonyl Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| CHEMSWORTH | +91-261-2397244 | New Delhi, India | 6707 | 30 | Inquiry |
| TCI Chemicals (India) Pvt. Ltd. | 1800 425 7889 | New Delhi, India | 6778 | 58 | Inquiry |
| Alfa Aesar | 1 800 209 7001 | Maharashtra, India | 6913 | 58 | Inquiry |
| Thermo Fisher Scientific India Private Limited | 08048372474Ext 168 | Delhi, India | 8 | 58 | Inquiry |
| Fuxin Pharmaceutical | +86-021-021-50872116 +8613122107989 | China | 10297 | 58 | Inquiry |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 | China | 49392 | 58 | Inquiry |
| City Chemical LLC | 2039322489 | United States | 168 | 58 | Inquiry |
| Shaanxi Dideu Medichem Co. Ltd | +86-29-81148696 +86-15536356810 | China | 3878 | 58 | Inquiry |
| Hebei Yanxi Chemical Co., Ltd. | +86-17531190177; +8617531190177 | China | 5873 | 58 | Inquiry |
Related articles
- Reactions of Cobalt carbonyl
- Dicobalt octacarbonyl is a metal compound with composition Co2(CO)8. This metal carbonyl is used as a reagent and catalyst in ....
- Feb 24,2022





