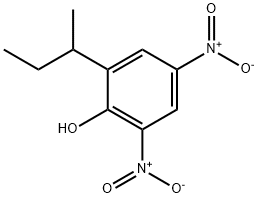
4,6-Dinitro-2-sec-butylphenol
- CAS No.
- 88-85-7
- Chemical Name:
- 4,6-Dinitro-2-sec-butylphenol
- Synonyms
- Dinoseb;DBNF;2-sec-Butyl-4,6-dinitrophenol;DNPB;Aatox;bnp20;bnp30;dn289;DNSBP;Dytop
- CBNumber:
- CB9497050
- Molecular Formula:
- C10H12N2O5
- Molecular Weight:
- 240.21
- MOL File:
- 88-85-7.mol
- Modify Date:
- 2024/3/14 15:18:32
| Melting point | 55.5°C |
|---|---|
| Boiling point | 382.92°C (rough estimate) |
| Density | 1.29 |
| vapor pressure | 0.01Pa at 25℃ |
| refractive index | 1.6620 (estimate) |
| Flash point | >100 °C |
| storage temp. | 2-8°C |
| solubility | Chloroform (Slightly), Methanol (Slightly) |
| pka | 4.62(at 25℃) |
| Specific Gravity | 1 |
| Water Solubility | 0.0052 g/100 mL |
| Merck | 13,3317 |
| Stability | Stable. Strong oxidizing agent. Incompatible with reducing agents, combustible material. |
| LogP | 1.26 at 22℃ |
| CAS DataBase Reference | 88-85-7(CAS DataBase Reference) |
| NIST Chemistry Reference | 2-Sec-butyl-4,6-dinitrophenol(88-85-7) |
| EPA Substance Registry System | Dinoseb (88-85-7) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |     GHS05,GHS06,GHS08,GHS09 |
|---|---|
| Signal word | Danger |
| Hazard statements | H300-H311-H315-H317-H318-H360-H410 |
| Precautionary statements | P201-P273-P280-P301+P310-P302+P352+P312-P305+P351+P338 |
| Hazard Codes | T;N,N,T,Xn |
| Risk Statements | 24/25-44-50/53-61-62-36-63-43-36/37/38-23/24/25-45-67-40-41-38 |
| Safety Statements | 45-53-60-61-36/37-24/25-23-26-36/37/39 |
| RIDADR | UN 2810 |
| WGK Germany | 3 |
| RTECS | SJ9800000 |
| HazardClass | 6.1(a) |
| PackingGroup | II |
| Toxicity | LD50 in adult male, female rats (mg/kg): 27, 28 orally (Gaines, Linder) |
4,6-Dinitro-2-sec-butylphenol price More Price(2)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 48378 | Dinoseb solution 200?μg/mL in methylene chloride, PESTANAL?, analytical standard | 88-85-7 | 1ML | ₹3085.6 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 45453 | Dinoseb PESTANAL?, analytical standard | 88-85-7 | 100MG | ₹4156.8 | 2022-06-14 | Buy |
4,6-Dinitro-2-sec-butylphenol Chemical Properties,Uses,Production
Description
Dinoseb, also known as dinitrophenol, can adversely affect the
energy generating reaction in a cell. No cell will live very long
under the influence of high concentrations of dinitrophenol. It
makes the body burn enough energy to result in weight loss.
During the 1930s, physicians unwittingly prescribed certain
types of dinitrophenol as diet pills.
Dow Chemical changed the basic structure of dinitrophenol
slightly to produce dinoseb, which was marketed in 1948.
Dinoseb was widely used as a contact herbicide against
broadleaf weeds.
Dinoseb causes toxicity the same way in plants,
animals, and fungi because all cells contain very similar
biochemical pathways for creating energy from the breakdown
of sugars. Furthermore, photosynthesis in plants
relies on an energy transfer system that is also inhibited by
dinitrophenol.
Given the high toxicity, EPA concluded that the doses
causing the birth defects and the endocrine-disrupting effects
were close to worker exposure levels. Thus, under an emergency
order issued in 1986, EPA suspended dinoseb’s registration.
In August 1990, the EPA banned the burying of
dinoseb-contaminated soils in EPA-approved landfills,
making incineration the only EPA-approved disposal method
for dinoseb-contaminated soil. Incineration is expensive and
incomplete, leaving a noncombustible residue as a further
hazardous waste and some combustion products that could
remain toxic. Therefore, an ex situ soil bioremediation process
was developed by the Sabre Processing company. This process
is known as the SABRE process; it uses an anaerobic consortium
and supplemental carbon source at the field scale to
successfully remediate contaminated soils.
Chemical Properties
solid
Uses
The amine, ammonium salt or acetate ester is used as a contact herbicide for postemergence weed control in cereals, cotton, peas, beans, potatoes, pumpkins, soybeans and strawberries.
General Description
Orange-brown viscous liquid or orange-brown solid. Orange crystals when pure. Has a pungent odor. Used as a plant growth regulator; insecticide and herbicide.
Reactivity Profile
4,6-Dinitro-2-sec-butylphenol is a powerful oxidizing agent. . 4,6-Dinitro-2-sec-butylphenol is dangerously explosive. When not water wet 4,6-Dinitro-2-sec-butylphenol is a high explosive. Dry, the material is easily ignited and 4,6-Dinitro-2-sec-butylphenol will burn very vigorously. On decomposition, nitro compounds such as this emit toxic fumes. Appear to be stable in acid solution, but are susceptible to decomposition by ultraviolet radiation in alkaline solution. [EPA, 1998].
Health Hazard
Extremely toxic: Probable oral lethal dose is 5-50 mg/kg; between 7 drops and 1 teaspoonful for 70 kg person (150 lb.).
Fire Hazard
This is a dinitrophenol herbicide. (Non-Specific -- Dinitrophenol, Flammable Solid). 4,6-Dinitro-2-sec-butylphenol is dangerously explosive. When not water wet 4,6-Dinitro-2-sec-butylphenol is a high explosive. Dry, the material is easily ignited and 4,6-Dinitro-2-sec-butylphenol will burn very vigorously. On decomposition, nitro compounds such as this emit toxic fumes. Appear to be stable in acid solution, but are susceptible to decomposition by ultraviolet radiation in alkaline solution.
Agricultural Uses
Plant growth regulator, Herbicide: Dinoseb is a phenolic herbicide used in soybeans, vegetables, fruits and nuts, citrus, and other field crops for the selective control of grass and broadleaf weeds (e.g., in corn). It is also used as an insecticide in grapes, and as a seed crop drying agent. It is produced in emuslifiable concentrates or as water-soluble ammonium or amine salts. It is no longer available in the U.S. Formerly widely used in the UK for the fumigation of potatoes; however, dinoseb acetate and dinoseb amine were banned from use in 1988. Dinoseb’s primary use is as a contact herbicide for post-emergence weed control in cereals, undersown cereals, seedling lucerne and peas. Dinoseb is also used as a corn yield enhancer and an insecticide and miticide. Banned for use in EU countries (includes salts and acetate). A U.S. EPA restricted Use Pesticide (RUP). The use of dinoseb was canceled in the U.S. in 1986 based on the potential risk of birth defects and other adverse health effects for applicators and other persons having substantial dinoseb exposure. There are 20 global suppliers.
Trade name
AATOX®; AI3-01122®; ARETIT®; BASANITE®; BNP 20®; BNP 30®; BUTAPHENE®; CALDON®; CASWELL No. 392DD®; CHEMOX®[C]; CHEMOX GENERAL®[C]; CHEMOX P. E. ®[C]; CHEMSECT DNBP®; DESICOIL®; DIBUTOX®; DINITRALL®; DINITRO®; DN 289®; DOW GENERAL®[C]; DOW GENERAL WEED KILLER®[C]; DOW SELECTIVE WEED KILLER®[C]; DYNAMYTE®[C]; DYTOP®; ELGETOL 318®; FANICIDE®; GEBUTOX®; HEL-FIRE®[C]; HIVERTOX®; HOE 26150®; IVOSIT®; KILOSEB®; KNOWX-WEED®; KNOX-WEED®; LADOB®; LASEB®; LIRO DNBP®; NITROPONE C®; PERSEVTOX®; PHENOTAN®; PREMERGE®; SINOX GENERAL®[C]; SPARIC®; SPURGE®; SUBITEX®; UNICROP DNBP®; VERTAC DINITRO WEED KILLER®[C]; VERTAC GENERAL WEED KILLER®[C]; VERTAC SELECTIVE WEED KILLER®[C]
Environmental Fate
Biological. When 14C-labeled dinoseb (5 ppm) was incubated in soil at 25°C for 60 days, 36.0% of the applied amount degraded to 14CO2 (Doyle et al., 1978). Thom and Agg (1975) reported that dinoseb is unlikely to be degraded in conventional sewage treatment processes.
Groundwater. According to the U.S. EPA (1986) dinoseb has a high potential to leach to groundwater.
Plant. When dinoseb on bean leaves was exposed to sunlight, photodegradation resulted in the formation of persistent, polar compounds. The compounds could not be identified by TLC (Matsuo and Casida, 1970).
Chemical/Physical. Reacts with organic and inorganic bases forming water-soluble salts (Worthing and Hance, 1991).
Emits toxic fumes of chlorine when heated to decomposition (Sax and Lewis, 1987).





