亜りん酸トリエチル 化学特性,用途語,生産方法
外観
無色澄明の液体
溶解性
水に難溶, エタノールに混和。エタノール及びアセトンに極めて溶けやすく、水にほとんど溶けない。
解説
C6H15O3P(166.16).(C2H5O)3P.亜リン酸トリエチル,トリエチルホスファイトともいう.三塩化リンとエタノールまたはエトキシドとを反応させると得られる.特有の臭気を有する無色の液体.沸点156 ℃,57~58 ℃(2.13 kPa).[α]20D 0.969.n20D 1.413.エタノール,エーテルに可溶,水に難溶.アルキル化試薬,脱酸素,脱硫黄試薬,殺虫剤合成中間体として使用される.高分子安定剤にも用いられる.森北出版「化学辞典(第2版)
用途
ホスホン酸エチルの合成やアルキル化試薬、脱酸素、脱硫黄試薬として有機合成に広く用いられる。高分子安定剤、難燃性高分子、潤滑油添加剤、殺虫剤などの合成原料。
化学的特性
Triethyl phosphite is a clear colorless liquid with a strong foul odor. Insoluble in water; soluble in alcohol and ether. Combustible.Vapors heavier than air.
使用
Triethyl phosphite is an organophosphorus compound. It is used as a reducing agent; can react with electrophiles to form phosphonates or phosphates; forms a stable complex with copper(I) iodide.
Triethyl phosphite is a very good nucleophile. The carbon adjacent to the bromine is the most electrophilic position, and phosphorus is the only nucleophile. Triethyl phosphite displaces the bromine in an SN2-like process, and back attack by the bromide which is released generates a phosphonate product, in which the α-protons are strongly acidic.
製造方法
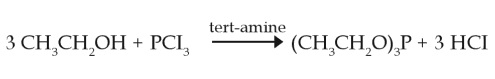
Triethyl phosphite is produced in a closed system by reaction of phosphorous trichlorid and ethanol in the presence of an inorganic or organic base. The product is purified by distillation (Bayer AG, 2002a).
Triethyl phosphite is exclusively used as an intermediate for the manufacturing of different products: flame retardants (about 60 %), optical brighteners (about 15 %), pesticides (about 15 %), antioxidants (about 5 %), and pharmaceuticals (about 5 %).
反応性
This reaction is currently used for the preparation of synthetically useful phosphonate reagents employed in modified retinal studies. Thus, diethyl 3-alkoxycarbonyl-2-propenylphosphonates are prepared in 72-91% yields by the reaction of methyl or ethyl 4-bromocrotonates with triethyl phosphite at 150-160°C. Similarly, diethyl 3-(ethoxycarbonyl)-2 methyl-2-propenylphosphonate is prepared in 81% yield from triethyl phosphite and ethyl 3-methyl-4-chlorocrotonate by heating at 180- 200°C.The diethyl (E)- and (Z)-3-ethoxycarbonyl-3 fluoro-2 -methyl-2-propenylphosphonates are respectively obtained from triethyl phosphite and (E)- or (Z)-4-bromo-2-fuoro-3-methyl-2-butenoates at 140°C.
反応プロフィール
Triethyl phosphite is colorless, moderately toxic liquid, combustible. Flammable when exposed to heat or flame. When heated to decomposition Triethyl phosphite emits toxic fumes of oxides of phosphorus [Lewis, 3rd ed., 1993, p. 1271].
健康ハザード
Exposure to high concentrations may cause headache, nausea, and dizziness due to reduced chlolinesterase activity.
火災危険
Special Hazards of Combustion Products: May form hazardous decomposition products.
安全性プロファイル
Moderately toxic by
ingestion. A skin and eye irritant. Flammable
liquid when exposed to heat, sparks, or
flame. When heated to decomposition it
emits toxic fumes of POx.
亜りん酸トリエチル 上流と下流の製品情報
原材料
準備製品
2-(2,3-DIHYDRO-1BENZENESULFONYL-PYRROLO[2,3-B]PYRIDIN-3-YL)ETHANAMINE
4'-(2-シアノスチリル)-4-スチルベンカルボニトリル
2-[2-(2-チエニル)エチル]安息香酸
(N-メトキシ-N-メチルカルバモイルメチル)ホスホン酸ジエチル
Phosphite
エチルジチオホスホン酸O-エチルS-フェニル
1,1-シクロペンタン二酢酸
2,1,3-ベンゾオキサジアゾール-4-カルボキシアルデヒド
2-アミノエチルホスホン酸
1,2-ジ(4-ピリジル)エチレン
(4-ニトロベンジル)ホスホン酸ジエチル
1-CYCLOPENTYLIDENE-PROPAN-2-ONE
スピロ[4.5]デカン-7,9-ジオンシクロペンタノン
9,10-ジヒドロ-4H-ベンゾ[4,5]シクロヘプタ[1,2-B]チオフェン-4-オン (90%)
4,4'-ビス(2-スルホナトスチリル)ビフェニル二ナトリウム
ホスカルネットナトリウム
2-メチルシクロプロパンカルボン酸
7-メチル-1,6-オクタジエン
N-(2-Methylpropylidene)-butylamine
2-(b-(2-Thienyl)vinyl)benzoicacid
2-(2,3-DIHYDRO-1-BENZENESULFONYL-PYRROLO[2,3-B]PYRIDIN-3-YL)ACETONITRILE
Optical Brighter FP127
2-テニルホスホン酸ジエチル
プロピルホスホン酸無水物
3,7-ジメチル-2,6-オクタジエン酸