ケテン 化学特性,用途語,生産方法
用途
有機合成原料
解説
母体となる化合物CH2=CO,C2H2O(42.04)をケテンとよぶ.実験室的にはアセトンの熱分解により,工業的には酢酸の熱分解でつくられる.常温で気体.
CH3COCH3 → CH2CO + CH4
CH3COOH → CH2CO + H2O
融点-151 ℃,沸点-56 ℃.2分子からジケテン(ケテンダイマー)が容易に生じる.ジケテンは熱分解によりケテンに戻る.
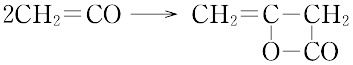
ケテンはヒドロキシ基,アミノ基,メルカプト基を有する化合物と反応してアセチル誘導体を与える.
化学的特性
Colorless gas with a sharp penetrating odor;liquefies at -56°C (-68.8°F); solidifies at-151°C (-239.8°F); soluble in alcohol andacetone, decomposed by water.
使用
For the conversion of higher acids into their anhydrides; for acetylation in the manufacture of cellulose acetate and aspirin.
調製方法
Ketene may be prepared also by pyrolysis of acetic anhydride or phenyl acetate or diketene. Other sources are quite unsatisfactory from a standpoint of yield. Small quantities may be made conveniently by heating acetone in a “ketene lamp.” This is a glass apparatus containing a Nichrome filament, heated electrically to red heat. Larger amounts are made by passing acetone or acetic acid through a tube at 700 °C. A very brief contact time is required, so that much of the acetone is undecomposed and has to be condensed and recycled. Also, it is imperative that the reaction tube be of inert material such as porcelain, glass, quartz, copper or stainless steel. A copper tube, if used, should be protected from oxidation by an iron sheath. Inert packing may be used (glass, vanadium pentoxide, porcelain), but just as good yields are obtained with empty tubes. No catalyst is known which accelerates this decomposition at significantly lower temperatures.
健康ハザード
Ketene is a highly toxic gas. It causes severeirritation to the eyes, nose, throat, and skin.Exposure to 10–15 ppm for several minutescan injure the respiratory tract. It causespulmonary edema. A 30-minute exposure to23 ppm was lethal to mice and a 10-minuteexposure to 200 and 750 ppm caused deathto monkeys and cats.
火災危険
Ketene in its gaseous state should be
flammable and explosive in air. The pure
compound, however, polymerizes readily
and cannot be stored as a gas. Its flash point
and LEL and UEL values are not reported. It
can react violently with oxidizers and many
organic compounds. Its small size and the
olefinic unsaturation impart further reactivity
to the molecule.
純化方法
Ketene is prepared by pyrolysis of acetic anhydride. Purify it by passing through a trap at -75o and collecting in a liquid-nitrogen-cooled trap. Ethylene is removed by evacuating the ethylene in an isopentane-liquid-nitrogen slush pack at -160o. Store it at room temperature in a suitable container in the dark or better at -80o, but do not store it under pressure as it may EXPLODE. It is a strong IRRITANT when inhaled and is as poisonous as phosgene. See diketene in “Heterocyclic Compounds”, Chapter 4. [Hurd Org Synth Coll Vol I 330 1941, Andreades & Carlson Org Synth Coll Vol V 679 1973.]
ケテン 上流と下流の製品情報
原材料
準備製品