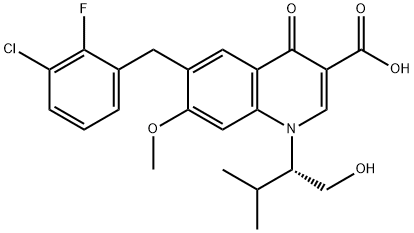
Elvitegravir
|
|
Elvitegravir 속성
- 녹는점
- 93-96°C
- 끓는 점
- 623.6±55.0 °C(Predicted)
- 밀도
- 1.357±0.06 g/cm3(Predicted)
- 저장 조건
- Refrigerator
- 용해도
- DMSO(약간 용해됨), 메탄올(약간 용해됨)
- 산도 계수 (pKa)
- 0.44±0.20(Predicted)
- 물리적 상태
- 고체
- 물리적 상태
- 단단한 모양
- 색상
- 황백색에서 연한 노란색
- InChIKey
- JUZYLCPPVHEVSV-LJQANCHMSA-N
- SMILES
- N1([C@H](CO)C(C)C)C2=C(C=C(CC3=CC=CC(Cl)=C3F)C(OC)=C2)C(=O)C(C(O)=O)=C1
- CAS 데이터베이스
- 697761-98-1
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 그림문자(GHS): | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 신호 어: | |||||||||||||||
| 유해·위험 문구: |
|
||||||||||||||
| 예방조치문구: |
|
Elvitegravir C화학적 특성, 용도, 생산
개요
In November 2013, the European Medicines Agency (EMA) approved elvitegravir (also known as GS 9137 and JTK 303) as a single agent to be used as part of an antiviral regimen that includes a ritonavir-boosted protease inhibitor for the treatment of HIV-1 in adults without mutations indicative of elvitegravir resistance. Elvitegravir is the second of three marketed HIV integrase strand transfer inhibitors (INSTIs) including raltegravir and dolutegravir (this volume of ARMC). Elvitegravir was discovered by modification of a literature naphthyridine HIV integrase inhibitor in which the naphthyridine core served as a bioisostere for the diketo acid moiety in an original series. Serendipitously, a 4-quinolone-3-carboxylic acid precursor en route to the desired bioisosteric glyoxylic acid demonstrated modest integrase inhibition (IC50=1600 nM). Further derivatization led to elvitegravir with enhanced inhibition of integrase strand transfer (IC50=7.2 nM) and significant antiviral activity (EC50=0.9 nM). Elvitegravir was prepared in seven synthetic steps from 2,4-difluoro-5-iodobenzoic acid. The corresponding acid chloride was coupled to ethyl 3-(dimethylamino) acrylate and further substituted with S-valinol. Base promoted cyclization afforded the quinolone which was protected as silyl ether. Negishi coupling installed the 2-fluoro-3-chlorobenzyl moiety. Subsequent hydrolysis and methoxylation afforded elvitegravir.화학적 성질
Off-White to Pale Yellow Solid용도
A novel inhibitor of human immunodeficiency virus type 1 integrase.정의
ChEBI: A quinolinemonocarboxylic acid that is 7-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid substited at position 1 by a 1-hydroxy-3-methylbutan-2-yl group and at position 6 by a 3-chloro-2-fluorobenzyl group (the S-enantiomer). It is us d in combination therapy for the treatment of HIV-1 infection.Elvitegravir 준비 용품 및 원자재
원자재
준비 용품
Elvitegravir 공급 업체
글로벌( 235)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Hangzhou Benoy Chemical Co., Ltd | +8617342059697 |
sales@benoychem.com | China | 315 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 |
sales@capotchem.com | China | 29798 | 60 |
| Beijing Cooperate Pharmaceutical Co.,Ltd | 010-60279497 |
sales01@cooperate-pharm.com | CHINA | 1811 | 55 |
| Hangzhou FandaChem Co.,Ltd. | 008657128800458; +8615858145714 |
fandachem@gmail.com | China | 9337 | 55 |
| career henan chemical co | +86-0371-86658258 +8613203830695 |
sales@coreychem.com | China | 29889 | 58 |
| Biochempartner | 0086-13720134139 |
candy@biochempartner.com | CHINA | 967 | 58 |
| HubeiwidelychemicaltechnologyCo.,Ltd | 18627774460 |
faith@widelychemical.com | CHINA | 742 | 58 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 |
linda@hubeijusheng.com | CHINA | 22968 | 58 |
| BOC Sciences | +1-631-485-4226 |
inquiry@bocsci.com | United States | 19553 | 58 |
| Chongqing Chemdad Co., Ltd | +86-023-6139-8061 +86-86-13650506873 |
sales@chemdad.com | China | 39916 | 58 |
Elvitegravir 관련 검색:
DELAVIRDINE
Orlistat
Tenofovir
Foscarnet sodium
Succinylcholine Chloride
Glutathione
ISOBUTANE
Tenofovir disoproxil fumarate
Emtricitabine
Sofosbuvir
Azelnidipine
4-[[4-[[4-[(E)-2-cyanoethenyl]-2,6-dimethyl-phenyl]amino]pyrimidin-2-yl]amino]benzonitrile
BMS707035
GSK1349572
Raltegravir
Etravirine
Atazanavir sulfate
4-OXO-1,4-DIHYDROQUINOLINE-3-CARBOXYLIC ACID