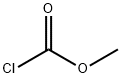
클로로탄산 메틸에스테르
|
|
클로로탄산 메틸에스테르 속성
- 녹는점
- -61°C
- 끓는 점
- 70-72 °C(lit.)
- 밀도
- 1.223 g/mL at 25 °C(lit.)
- 증기 밀도
- 3.26 (vs air)
- 증기압
- 4.8 psi ( 20 °C)
- 굴절률
- n
20/D 1.387(lit.)
- 인화점
- 64 °F
- 저장 조건
- Store at 0-5°C
- 용해도
- 클로로포름, 에틸아세테이트에 용해됨
- 물리적 상태
- 액체
- 색상
- 무색투명~연황색
- 냄새
- 매운.
- 수용성
- 가수 분해
- Merck
- 13,6071
- Dielectric constant
- 12.9
- 안정성
- 수분에 민감한
- CAS 데이터베이스
- 79-22-1(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | F,T+ | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 11-21/22-26-34 | ||
| 안전지침서 | 14-26-28-36/37/39-45-63-46-28A-39-36/37 | ||
| 유엔번호(UN No.) | UN 1238 6.1/PG 1 | ||
| WGK 독일 | 2 | ||
| RTECS 번호 | FG3675000 | ||
| F 고인화성물질 | 10-19-21 | ||
| 자연 발화 온도 | 485 °C | ||
| 위험 등급 | 6.1(a) | ||
| 포장분류 | I | ||
| HS 번호 | 29151300 | ||
| 유해 물질 데이터 | 79-22-1(Hazardous Substances Data) | ||
| 기존화학 물질 | KE-04746 |
클로로탄산 메틸에스테르 C화학적 특성, 용도, 생산
화학적 성질
Methyl chloroformate is a colorless liquid with an unpleasant, acrid odor. This is a highly corrosive and flammable material. Decomposed by hot water; stable to cold water. Soluble in methanol alcohol, ether, and benzene.용도
Chloroformic Acid Methyl Ester is used in the synthesis of a new class of potent Cdk4 inhibitors in the treatment of cancer. Also used in the synthesis of Phorboxazole B.주요 응용
Methyl chloroformate (MCF) is generally used for the derivatization of functional groups such as carboxylic acids, amines, and phenols.MCF can also be used:
To activate 3-acylpyridines for nucleophilic addition with alkynyltin reagents to form 2,3-disubstituted 1,2-dihydropyridines.
Chloroesterification of terminal alkynes to form β-chloro-α,β-unsaturated esters.
As an electrophilic reagent to mediate the reaction of pyridine with lithium dialkyl- or diarylcuprates to form 4-substituted 1,4-dihydropyridine derivatives.
To convert nitronates into methoxycarbonyl nitronates.
생산 방법
Prepared from phosgene and methyl alcohol.일반 설명
A colorless liquid with a pungent odor. Flash point 54°F. Corrosive to metals and tissue. Vapors heavier than air. Very toxic by inhalation. Used to make other chemicals and insecticides.공기와 물의 반응
Highly flammable. Gives off hydrochloric acid fumes in contact with moist air. Slightly soluble in water and decomposed by water to hydrochloric acid with evolution of heat.반응 프로필
Methyl chloroformate is incompatible with water, strong oxidizing agents, alcohols, bases (including amines). Decomposes slowly in water to yield methanol, HCl, and CO2; reaction can be hazardous if water is hot. Attacks many metals especially in humid atmosphere [Handling Chemicals Safely 1980. p. 476]. May react vigorously or explosively if mixed with diisopropyl ether or other ethers in the presence of trace amounts of metal salts [J. Haz. Mat., 1981, 4, 291].위험도
Flammable, dangerous fire risk. Highly corrosive and irritant to skin and eyes.건강위험
Methyl chloroformate is highly toxic upon inhalation and upon ingestion. A concentration of 1 mg/liter (190 ppm) has been lethal in 10 minutes. It is corrosive and irritating to skin.화재위험
Methyl chloroformate is very dangerous when exposed to heat sources, sparks, flame, or oxidizers. Methyl chloroformate will react with water or steam to produce toxic and corrosive fumes. Vapors may travel to a source of ignition and flash back. Withdraw immediately in case of rising sound from venting safety device or any discoloration of tank due to fire. Toxic fumes of phosgene are produced when the material is heated to decomposition. Heat or steam should be avoided.화학 반응
Reactivity with Water: Reacts slowly, evolving hydrogen chloride (hydrochloric acid). Reaction can be hazardous if water is hot; Reactivity with Common Materials: Corrodes rubber; Stability During Transport: Stable; Neutralizing Agents for Acids and Caustics: Flush with water, rinse with sodium bicarbonate or lime solution; Polymerization: Not pertinent; Inhibitor of Polymerization: Not pertinent.Safety Profile
Poison by ingestion, inhalation, and intraperitoneal routes. Moderately toxic by skin contact. Human systemic effects by inhalation: conjunctiva irritation and respiratory effects. Corrosive to skin, eyes, and mucous membranes. Very dangerous fire hazard when exposed to heat sources, sparks, flame, or oxidzers. Reacts with water or steam to produce toxic and corrosive fumes. When heated to decomposition it emits toxic fumes of Cl-, methyl chloroformate, and phosgene.잠재적 노출
Used in synthesis of pharmaceuticals; herbicides, plastics and other organic chemicals; as a solvent in the photographic industry; as a chemical intermediate in the production of other chemicals. In WWI it was used as military tear-producing warfare agent.운송 방법
UN1238 Methyl chloroformate, Hazard class: 6.1; Labels: 6.1-Poison Inhalation Hazard, 3-Flammable liquid, 8-Corrosive material Inhalation Hazard Zone A비 호환성
May form explosive mixture with air. Violent reaction with alkali metals; ethers. Incompatible with strong acids; strong bases; alcohols, oxidizers, dimethylsulfoxide; dimethyl formamide. Contact with water or moisture produces corrosive and poisonous hydrogen chloride gas, methanol and carbon monoxide. Corrodes metals in the presence of moisture. Attacks some plastics, rubber and coatings.폐기물 처리
Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste containing this contaminant (≥100 kg/mo) must conform to EPA regulations governing storage, transportation, treatment, and waste disposal클로로탄산 메틸에스테르 준비 용품 및 원자재
원자재
준비 용품
메틸시아노카바메이트
Oxibendazole
메톡사디아존
Cefaclor monohydrate
METHYL 3-THIOPHENECARBOXYLATE
PIPERAZINE-1,4-DICARBOXYLIC ACID TERT-BUTYL ESTER METHYL ESTER
FOSAMINE AMMONIUM
메틸 카바메이트
Tenidap
(2S)-2-amino-3-(5-bromo-1H-indol-3-yl)propanoic acid
Asulam
메틸티오시아노포르메이트
카벤다짐
5-메틸-L-트립토판
METHYL 3-BROMOTHIOPHENE-2-CARBOXYLATE
카보닐산알릴메틸에스테르
티오판산-메틸
SWEP
Phenmedipham
Mebendazole
Toloxatone
디메틸 카보네이트
N-METHOXYCARBONYLMALEIMIDE
보론