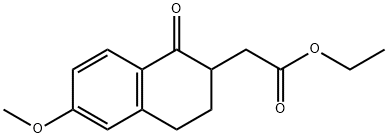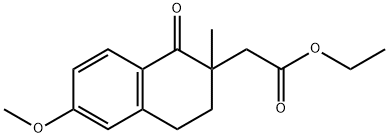
Ethyl 2-(6-methoxy-2-methyl-1-oxo-1,2,3,4-tetrahydronaphthalen-2-yl)acetate synthesis
- Product Name:Ethyl 2-(6-methoxy-2-methyl-1-oxo-1,2,3,4-tetrahydronaphthalen-2-yl)acetate
- CAS Number:1414797-83-3
- Molecular formula:C16H20O4
- Molecular Weight:276.33
Yield:1414797-83-3 81%
Reaction Conditions:
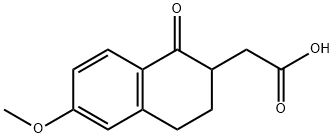
Stage #1: 6-methoxy-1,2,3,4-tetrahydro-1-oxonaphthalene-2-acetic acid, ethyl esterwith sodium hydride in N,N-dimethyl-formamide; for 0.666667 h;Cooling with ice;
Stage #2: methyl iodide in N,N-dimethyl-formamide at 20; for 6 h;
Steps:
3 Ethyl 2-(6-methoxy-2-methyl-l-oxo-l,2,3,4-tetrahvdronaphthalen-2-yl) acetate (3A):
Ethyl 2-(6-methoxy-2-methyl-l-oxo-l,2,3,4-tetrahvdronaphthalen-2-yl) acetate (3A): A solution of 1C (30 g, 1 14 mmol) in DMF (100 mL) was added over a period of 30 min to an ice cold solution of NaH (1 1.44 g, 286 mmol) in DMF (100 mL), and the mixture was stirred for 10 min. Methyl iodide (21.36 mL, 343 mmol) was then added, and the mixture was stirred for 6 h at room temperature. The reaction was then brought to 0 °C, excess NaH was quenched with ice water, and the aqueous layer was extracted with ethyl acetate (2 x 200 mL). The combined organic layers were dried over sodium sulfate and filtered, and the filtrate was concentrated under vacuum to give the title compound (30 g, 81%) as a solid. H NMR (400 MHz, DMSO-6): δ 7.96 (d, J= 8.4 Hz, 2H), 6.72 (dd, J= 2.4 Hz, J2 = 8.4 Hz, 1H), 6.62 (s, 1H), 4.09 (q, J= 7.2 Hz, 2H), 3.08 - 2.80 (m, 3H), 2.46 (d, J= 15.6 Hz, 1H), 2.39 (dd, J; = 4.4 Hz, J2 = 12.0 Hz, 1H), 1.90 (dt, J = 14 Hz, J2 = 4.4 Hz, 1H), 1.26 (s, 3H), 1.21 (t, J= 7.2 Hz, 1H). ESI-MS m/z: 277 (M+H)+.
References:
WO2014/74761,2014,A2 Location in patent:Page/Page column 35
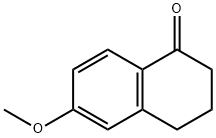
1078-19-9
368 suppliers
$6.00/5g

1414797-83-3
1 suppliers
inquiry
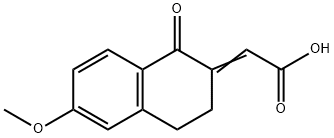
13587-70-7
3 suppliers
inquiry

1414797-83-3
1 suppliers
inquiry
17529-16-7
10 suppliers
inquiry

1414797-83-3
1 suppliers
inquiry