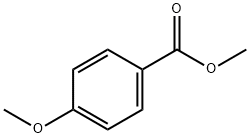
Methyl anisate synthesis
- Product Name:Methyl anisate
- CAS Number:121-98-2
- Molecular formula:C9H10O3
- Molecular Weight:166.17

123-11-5
839 suppliers
$10.00/10g

74-88-4
341 suppliers
$15.00/10g

121-98-2
280 suppliers
$12.00/1mg
Yield:121-98-2 84%
Reaction Conditions:
with tert.-butylhydroperoxide;sodium azide;tetra-(n-butyl)ammonium iodide in toluene at 90; for 2 h;
Steps:
2 Example 2: Preparation of methyl 4-methoxybenzoate
Take a clean 15mL high pressure reaction bottle,Was added p-methoxybenzaldehyde (74.8 mg, 0.55 mmol), tetrabutylammonium iodide (40.6 mg, 0.11 mmol)Sodium azide (107.25 mg, 1.65 mmol)Oxidant tert-butyl hydroperoxide (0.10 mL, 0.825 mmol)And methyl iodide (156.15 mg, 1.1 mmol)Solvent Toluene (1.0 mL),Add a small magnet,90 reaction 2h,Dichloromethane (3 x 10 mL). The organic phase was decompressed under reduced pressure and extracted with ethyl acetate: petroleum ether = 1: 30 solvent as eluant, 300 column chromatography silica gel as filler,The title product was purified by column chromatography (76.71 mg, yield 84%).
References:
Hangzhou Normal University;Zhong Guofu;Zeng Xiaofei;Chen Qiliang;Shen Dan;Ding Liyuan CN106588644, 2017, A Location in patent:Paragraph 0049; 0050; 0051; 0126; 0127; 0128

67-56-1
737 suppliers
$7.29/5ml-f
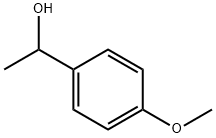
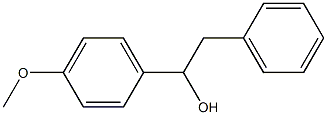
3319-15-1
106 suppliers
$10.00/1g

121-98-2
280 suppliers
$12.00/1mg

67-56-1
737 suppliers
$7.29/5ml-f
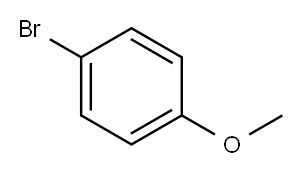
104-92-7
424 suppliers
$10.00/10g
201230-82-2
1 suppliers
inquiry

121-98-2
280 suppliers
$12.00/1mg
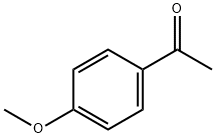
100-06-1
575 suppliers
$18.19/25g

121-98-2
280 suppliers
$12.00/1mg