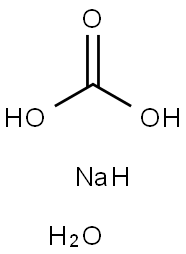
SODIUM CARBONATE, MONOHYDRATE
- CAS No.
- 5968-11-6
- Chemical Name:
- SODIUM CARBONATE, MONOHYDRATE
- Synonyms
- NA2CO3;SODA ASH;SODA;DENSE SODA ASH;SODIUM CARBONATE DENSE;DENSE SODIUM CARBONATE;SODIUM CARBONATE-1-HYDRATE;SOLVAY SODA;Soda crystals;SOD.CARBONATE
- CBNumber:
- CB1704053
- Molecular Formula:
- CH5NaO4
- Molecular Weight:
- 104.04
- MOL File:
- 5968-11-6.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/3/7 16:15:27
| Melting point | 851 °C(lit.) |
|---|---|
| Boiling point | 1600 °C |
| Density | 2.53 |
| storage temp. | Store at RT. |
| solubility | H2O: 1 M at 20 °C, clear, colorless |
| form | Powder/Solid |
| Specific Gravity | 2.25 |
| color | White |
| Water Solubility | Soluble in water. Insoluble in alcoholSoluble in water and glycerol. Insoluble in alcohol, acetone, alkyl acetates,benzonitrile and liquid ammonia. |
| Sensitive | Hygroscopic |
| Merck | 14,8596 |
| Dielectric constant | 3.0(Ambient) |
| Stability | Stable. Incompatible with strong acids, aluminium, fluorine, lithium, phosphorus oxides. |
| LogP | -0.809 (est) |
| CAS DataBase Reference | 5968-11-6(CAS DataBase Reference) |
| EPA Substance Registry System | Sodium carbonate, monohydrate (5968-11-6) |
SODIUM CARBONATE, MONOHYDRATE price More Price(17)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | S4132 | Sodium carbonate monohydrate ReagentPlus?, ≥99.5% | 5968-11-6 | 500G | ₹5683.13 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 230952 | Sodium carbonate monohydrate ACS reagent, ≥99.5% | 5968-11-6 | 100G | ₹4243.4 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | S4132 | Sodium carbonate monohydrate ReagentPlus?, ≥99.5% | 5968-11-6 | 1KG | ₹8281.13 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 230952 | Sodium carbonate monohydrate ACS reagent, ≥99.5% | 5968-11-6 | 500G | ₹7555.85 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 230952 | Sodium carbonate monohydrate ACS reagent, ≥99.5% | 5968-11-6 | 2.5KG | ₹11647.7 | 2022-06-14 | Buy |
SODIUM CARBONATE, MONOHYDRATE Chemical Properties,Uses,Production
Chemical Properties
white powder
Uses
Buffers; pH adjustment.
Production Methods
Sodium carbonate is produced by the ammonia-soda process, also known as the Solvay process.
Pharmaceutical Applications
Sodium carbonate is used as an alkalizing agent in injectable,
ophthalmic, oral, and rectal formulations.
In effervescent tablets or granules, sodium carbonate is used in
combination with an acid, typically citric acid or tartaric acid.
When the tablets or granules come into contact with water, an acid–
base reaction occurs in which carbon dioxide gas is produced and
the product disintegrates. Raw materials with low moisture
contents are required to prevent the early triggering of the
effervescent reaction.
As an alkalizing agent, concentrations of sodium carbonate
between 2% and 5% w/w are used in compressed tablet
formulations. As an effervescent agent, concentrations of
sodium carbonate up to 10% w/w can be used.
Therapeutically, sodium carbonate is also used as an oral
antacid.
Industrial uses
The most prominent characteristic of soda ash in solution is the high buffered pH response. For example, changing the sodium carbonate dosage from 0.03% to 30% only causes the solution pH to change from 10.5 to 11.7. Because of this, in mineral processing application, soda ash is used for pH control to a maximum value of 10.5.
Safety
Sodium carbonate is used in injectable, oral, and rectal pharmaceutical
formulations. The pure form of sodium carbonate is mildly
toxic by ingestion, moderately toxic by inhalation and SC routes,
and very toxic by the IP route. It is irritating to the skin and eyes.
Dust and vapors of sodium carbonate may irritate mucous
membranes, causing coughing and shortness of breath. It also has
experimental reproductive effects.
Sodium carbonate can migrate to food from packaging
materials. When used as an excipient or antacid, sodium carbonate
is generally regarded as a nontoxic and nonirritating material.
LD50 (mouse, IP): 0.12 g/kg
LD50 (mouse, SC): 2.21 g/kg
LD50 (rat, oral): 4.09 g/kg
storage
Sodium carbonate converts to the monohydrate form when in contact with water and produces heat. It begins to lose carbon dioxide at temperatures above 400℃ and decomposes before boiling. Store in airtight containers.
Incompatibilities
Sodium carbonate decomposes when in contact with acids in the presence of water to produce carbon dioxide and effervescence. It may react violently with aluminum, phosphorous pentoxide, sulfuric acid, fluorine, and lithium.
Regulatory Status
GRAS listed. Accepted for use as a food additive in Europe.
Included in the FDA Inactive Ingredients Database (injections;
ophthalmic solution; oral capsules and tablets; rectal suspensions).
Included in the Canadian List of Acceptable Non-medicinal
Ingredients. Included in parenteral (powder for solution for
injection) and nonparenteral medicines (oral effervescent tablets,
soluble tablets, granules, lozenges, chewing gums) licensed in the
UK.
USP32–NF27 allows either the anhydrous or the monohydrate
form.
SODIUM CARBONATE, MONOHYDRATE Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| LABDHI CHEMICAL INDUSTRIES | +91-9322229293 +91-9322229293 | Mumbai, India | 21 | 58 | Inquiry |
| Rishi Chemical Works Pvt Ltd | +91-3322290068 +91-3322290071 | Kolkata, India | 43 | 58 | Inquiry |
| oswal chemicals | +91-9173644055 +91-9173644055 | Gujarat, India | 66 | 58 | Inquiry |
| Nyne Organics Pvt Ltd | +91-9920180386 +91-9920180386 | Mumbai, India | 53 | 58 | Inquiry |
| Kronox Lab Sciences Pvt Ltd | +91-9313231074 +91-9313231074 | Gujarat, India | 240 | 58 | Inquiry |
| Merck Ltd | +91-2262109800 +91-2262109000 | Maharashtra, India | 272 | 58 | Inquiry |
| Sujata Chemicals | +91-9898025496 +91-9898025496 | Gujarat, India | 73 | 58 | Inquiry |
| Choice Chemicals | +91-9895031268 +91-9895031268 | Kerela, India | 118 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| EMCURE PHARMACEUTICALS LTD | +91 22 3061 0000 | New Delhi, India | 93 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| LABDHI CHEMICAL INDUSTRIES | 58 |
| Rishi Chemical Works Pvt Ltd | 58 |
| oswal chemicals | 58 |
| Nyne Organics Pvt Ltd | 58 |
| Kronox Lab Sciences Pvt Ltd | 58 |
| Merck Ltd | 58 |
| Sujata Chemicals | 58 |
| Choice Chemicals | 58 |
| JSK Chemicals | 58 |
| EMCURE PHARMACEUTICALS LTD | 58 |
Related articles
- How do you make soda ash?
- Soda ash is a safe and simple natural compound with white granular appearance and is a key ingredient in various industrial pr....
- Mar 7,2024
- Transformation between sodium carbonate monohydrate and sodium carbonate
- Sodium carbonate monohydrate can be obtained from the aqueous solution of the sodium carbonate by atmospheric evaporative crys....
- Feb 27,2020






