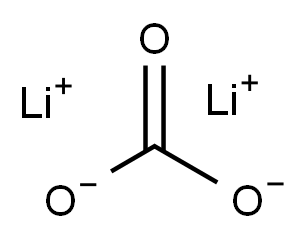
Lithium carbonate
- CAS No.
- 554-13-2
- Chemical Name:
- Lithium carbonate
- Synonyms
- Li2CO3;Eskalith;Carbolith;teralithe;carbolite;LITHIUM CARBONATE PURE;Lithium carbonate, TraceSelect;Limas;Phasal;Plenur
- CBNumber:
- CB5852228
- Molecular Formula:
- CLi2O3
- Molecular Weight:
- 73.89
- MOL File:
- 554-13-2.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/6/19 22:55:17
| Melting point | 720 °C |
|---|---|
| Boiling point | 1342 °C(lit.) |
| Density | 2.11 g/mL at 25 °C |
| Flash point | 1310°C |
| storage temp. | Store at +5°C to +30°C. |
| solubility | 13g/l |
| form | wire |
| pka | pKa 6.38 (Uncertain);10.25 (Uncertain) |
| Specific Gravity | 2.11 |
| color | White |
| PH | 10-11 (5g/l, H2O, 20℃) |
| Odor | odorless |
| Water Solubility | 13 g/L (20 ºC) |
| Merck | 14,5527 |
| Solubility Product Constant (Ksp) | pKsp: 1.6 |
| BRN | 3999191 |
| BCS Class | 1 |
| InChIKey | XGZVUEUWXADBQD-UHFFFAOYSA-L |
| LogP | -0.809 (est) |
| CAS DataBase Reference | 554-13-2(CAS DataBase Reference) |
| NIST Chemistry Reference | Lithium carbonate(554-13-2) |
| EPA Substance Registry System | Lithium carbonate (554-13-2) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H302-H319 | |||||||||
| Precautionary statements | P264-P270-P280-P301+P312-P305+P351+P338-P337+P313 | |||||||||
| Hazard Codes | Xn,C,F | |||||||||
| Risk Statements | 36/38-41-36/37/38-22-36-34-20/21/22-15-14-11 | |||||||||
| Safety Statements | 8-43-45-37/39-26-36/37-24/25-36/37/39-16-7/8-3/7/9 | |||||||||
| WGK Germany | 2 | |||||||||
| RTECS | OJ5800000 | |||||||||
| F | 10 | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 28369100 | |||||||||
| Toxicity | LD50 orally in rats: 0.71 g/kg (Smyth) | |||||||||
| NFPA 704 |
|
Lithium carbonate price More Price(43)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 62470 | Lithium carbonate puriss. p.a., ACS reagent, reagent (for microscopy), ≥99.0% (T) | 554-13-2 | 100G | ₹4059.38 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 752843 | Lithium carbonate 99.999% trace metals basis | 554-13-2 | 25G | ₹7956.38 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 62470 | Lithium carbonate puriss. p.a., ACS reagent, reagent (for microscopy), ≥99.0% (T) | 554-13-2 | 500G | ₹11398.73 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 431559 | Lithium carbonate 99.99% trace metals basis | 554-13-2 | 50G | ₹13563.73 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 255823 | Lithium carbonate ACS reagent, ≥99.0% | 554-13-2 | 100G | ₹2403.15 | 2022-06-14 | Buy |
Lithium carbonate Chemical Properties,Uses,Production
Chemical Properties
Lithium carbonate is a white hygroscopic powder.
Physical properties
White monoclinic crystals; refractive index 1.428; density 2.11 g/cm3; melts at 723°C; decomposes at 1,310°C; low solubility in water (1.54 g/100g) at 0°C; 1.32 g//100g at 20°C), solubility decrease with temperature (0.72g/100g at 100°C); insoluble in acetone and ethanol.
Uses
Lithium carbonate is used as a compound for producing metallic lithium. Lithium carbonate is the result of treating the mineral spodumene with sulfuric acid and then adding calcium carbonate. It is used as an antidepressant.
Preparation
Lithium carbonate is prepared by the precipitation of lithium ion by carbonate ion from an aqueous solution. Still another process, which is carried out on a smaller scale, is the reaction of a solution of lithium hydroxide with carbon dioxide gas. Lithium carbonate precipitates and is recovered from the supernatant solution.
Indications
Lithium inhibits thyroidal incorporation of I- into Tg, as well as the secretion of thyroid hormones, but it does not inhibit the activity of the Na+-I- symporter or the accumulation of I- within the thyroid. Lithium offers no particular advantage over drugs of the thionamide class but may be employed for temporary control of thyrotoxicosis in patients who are allergic to both thionamides and iodide.
Definition
lithium carbonate: A white solid,Li2CO3; r.d. 2.11; m.p. 723°C; decomposesabove 1310°C. It is producedcommercially by treating the ore with sulphuric acid at 250°C andleaching the product to give a solutionof lithium sulphate. The carbonateis then obtained by precipitationwith sodium carbonate solution.Lithium carbonate is used in the preventionand treatment of manicdepressivedisorders. It is also usedindustrially in ceramic glazes.
General Description
Lithiumcarbonate (Eskalith, Lithane) and lithium citrate(Cibalith-S) are the salts commercially available in theUnited States.
Reactivity Profile
A base. Decomposed by acids with the evolution of carbon dioxide. Fluorine burns fiercely on contact with Lithium carbonate.
Side effects
Drowsiness, dizziness, tiredness, increased thirst, increased frequency of urination, weight gain, and mildly shaking hands (fine tremor) may occur. These should go away as your body adjusts to the medication. This medication may increase serotonin and rarely cause a very serious condition called serotonin syndrome/toxicity. The risk increases if you are also taking other drugs that increase serotonin, so tell your doctor or pharmacist of all the drugs you take (see Drug Interactions section). Get medical help right away if you develop some of the following symptoms: fast heartbeat, hallucinations, loss of coordination, severe dizziness, severe nausea/vomiting/diarrhea, twitching muscles, unexplained fever, unusual agitation/restlessness.
Safety Profile
Human carcinogenic data. Poison by intraperitoneal and intravenous routes. Moderately toxic by ingestion and subcutaneous routes. Human systemic effects by ingestion: toxic psychosis, tremors, changes in fluid intake, muscle weakness, increased urine volume, nausea or vomiting, allergic dermatitis. Human reproductive effects by ingestion: effects on newborn, including Apgar score changes and other neonatal measures or effects. Human teratogenic effects by ingestion: developmental abnormalities of the cardiovascular system, central nervous system, musculoskeletal and gastrointestinal systems. An experimental teratogen. Experimental reproductive effects. Experimental carcinogen producing leukemia and thyroid tumors. Human mutation data reported. Used in the treatment of manic-depressive psychoses. Incompatible with fluorine. See also LITHIUM COMPOUNDS.
Potential Exposure
Lithium carbonate is used in treatment of manic-depressive psychoses; to make ceramics and porcelain glaze; varnishes, dyes, pharmaceuticals, coating of arc-welding electrodes; battery alloys; nucleonics, luminescent paints; glass ceramics; lubricating greases; in aluminum production
Shipping
UN2811 Toxic solids, organic, n.o.s., Hazard Class: 6.1; Labels: 6.1-Poisonous materials, Technical Name Required
Purification Methods
Crystallise it from water. Its solubility decreases as the temperature is raised. The solubility in H2O is 1.3% at ~10o, and 0.7% at ~100o. [D.nges in Handbook of Preparative Inorganic Chemistry (Ed. Brauer) Academic Press Vol I p 987 1963, Caley & Elving Inorg Synth I 1 1939.]
Incompatibilities
The aqueous solution is a strong base. Reacts violently with acids, powdered calcium and fluorine.Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong acids, powdered calcium, fluorine, moisture. Corrodes aluminum, copper, zinc.
Lithium carbonate Preparation Products And Raw materials
Raw materials
1of2
chevron_rightPreparation Products
1of4
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| PACIFIC ORGANICS PVT LTD | +91 9322467890 | Maharashtra, India | 51 | 58 | Inquiry |
| ULTIMA CHEMICALS | +91 7208029458 | Mumbai, India | 95 | 58 | Inquiry |
| Yogi Dye Chem Industries | +91-9821098577 +91-9821098577 | Mumbai, India | 124 | 58 | Inquiry |
| Global Pharma | +91-9819994077 +91-9819994077 | Maharashtra, India | 34 | 58 | Inquiry |
| BEE CHEMS | +91-9918601353 +91-8604471160 | Uttar Pradesh, India | 24 | 58 | Inquiry |
| SNECOFRi Pvt Ltd | +91-9032850129 +91-9032850129 | Telangana, India | 404 | 58 | Inquiry |
| VITRAG CHEMICALS PVT LTD | +91-7490053194 +91-7490053194 | Gujarat, India | 47 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| PANCHSHEEL ORGANICS LTD | +91-9821053955 +91-9324201019 | Mumbai, India | 82 | 58 | Inquiry |
| Kronox Lab Sciences Pvt Ltd | +91-9313231074 +91-9313231074 | Gujarat, India | 240 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| PACIFIC ORGANICS PVT LTD | 58 |
| ULTIMA CHEMICALS | 58 |
| Yogi Dye Chem Industries | 58 |
| Global Pharma | 58 |
| BEE CHEMS | 58 |
| SNECOFRi Pvt Ltd | 58 |
| VITRAG CHEMICALS PVT LTD | 58 |
| JSK Chemicals | 58 |
| PANCHSHEEL ORGANICS LTD | 58 |
| Kronox Lab Sciences Pvt Ltd | 58 |
Related articles
- Different applications of lithium carbonate
- Lithium carbonate(Li2CO3) is an inorganic compound.
- Jun 15,2022
554-13-2(Lithium carbonate)Related Search:
1of4
chevron_right




