Strontium carbonate
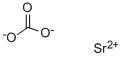
- CAS No.
- 1633-05-2
- Chemical Name:
- Strontium carbonate
- Synonyms
- STRONTIUM CARBONATE POWDER;SrCO;ci77837;Nsc112224;strontianite;Strontiumcarbonat;Sulfuryl caprylate;StrontuM carbonate;STRONTIUM CARBONAE;StorntiumCarbonate
- CBNumber:
- CB1237917
- Molecular Formula:
- CO3Sr
- Molecular Weight:
- 147.63
- MOL File:
- 1633-05-2.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/7/4 9:18:11
| Melting point | 1494 °C (lit.) |
|---|---|
| Density | 3.7 g/mL at 25 °C (lit.) |
| storage temp. | Inert atmosphere,Room Temperature |
| solubility | dilute aqueous acid: slightly soluble(lit.) |
| form | Powder |
| Specific Gravity | 3.7 |
| color | white |
| Water Solubility | Soluble in ammonium chloride. Slightly soluble in ammonia and water. |
| Merck | 14,8838 |
| Solubility Product Constant (Ksp) | pKsp: 9.25 |
| Stability | Stable. Incompatible with strong acids. |
| InChIKey | LEDMRZGFZIAGGB-UHFFFAOYSA-L |
| LogP | -0.809 (est) |
| CAS DataBase Reference | 1633-05-2(CAS DataBase Reference) |
| EPA Substance Registry System | Carbonic acid, strontium salt (1:1) (1633-05-2) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H315-H319-H335 | |||||||||
| Precautionary statements | P280-P302+P352-P304+P340 | |||||||||
| WGK Germany | - | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 2836920000 | |||||||||
| NFPA 704 |
|
Strontium carbonate price More Price(33)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 472018 | Strontium carbonate ≥99.9% trace metals basis | 1633-05-2 | 100G | ₹6646.55 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 472018 | Strontium carbonate ≥99.9% trace metals basis | 1633-05-2 | 500G | ₹21909.8 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 289833 | Strontium carbonate ≥98% | 1633-05-2 | 250G | ₹3150.08 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 204455 | Strontium carbonate 99.995% trace metals basis | 1633-05-2 | 5G | ₹3875.35 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 289833 | Strontium carbonate ≥98% | 1633-05-2 | 1KG | ₹4546.5 | 2022-06-14 | Buy |
Strontium carbonate Chemical Properties,Uses,Production
Description
Strontium carbonate has the formula of SrCO3 and the molecular weight of 147.6326 g/mol. Strontium carbonate occurs in nature as the mineral “strontianite”. The name strontianite comes from a famous location for the mineral, Strontian, Scotland. Strontianite is strontium carbonate as found naturally. It occurs as white or slightly gray orthorhombic crystals with a refractive index of 1.518. The unit-cell parameters are: a = 5.107 ? , b = 8.414 ? , c = 6.029 ? , Z = 4; V = 259.07 ? 3, Den(Calc) = 3.78. The crystal system is orthorhombic with space group Pmcn and point group 2/m, 2/m, 2/m. Strontium carbonate has only one stable form (aragonite-type structure) and temperature of precipitation has no effect on crystal form, unlike that of calcium or magnesium carbonates.
Chemical Properties
Strontium carbonate is a milky white free flowing powder. It is little more insoluble (Ksol=10-8.8) than calcium carbonate (Ksol=10-8.07), so it should not be surprising that under appropriate conditions Sr2+ can be precipitated by biogenic carbonate.
Chemical Properties
Heating strontium carbonate up from 1000°C to 1300°C causes the compound to undergo a decomposition reaction which results in the capture of thermal energy, shown here as the peak in the black curve occurring between Time = 20 and Time = 40 minutes. Cooling the system back down to 1000°C leads to reconstitution of the starting material in a carbonation reaction thereby releasing the stored thermal energy, a process shown as the valley in the black curve between Time = 50 and Time = 80 minutes.
Physical properties
White orthorhombic crystals; refractive index 1.518; hygroscopic; hardness 3.5 Mohs; density 3.5 g/cm3; insoluble in water; soluble in dilute acids with liberation of carbon dioxide.
Occurrence
Strontium carbonate occurs in nature as mineral strontianite. The compound is used in pyrotechnics and ceramic ferrites. It also is used in making iridescent glass for color television tubes. Other uses are in refining sugar and preparing other strontium salts.
Uses
Strontium carbonate (SrCO3) is used to make radiation-resistant glass and TV picture tubes, as well as pyrotechnics.
Preparation
Strontium carbonate occurs in nature as strontianite and can be mined from its deposit. It is, however, usually made commercially from the mineral “celestite”. Celestite is fused with sodium carbonate at elevated temperatures or boiled with a solution of ammonium carbonate.Strontium carbonate is insoluble in water. It precipitates from the product mixture in the second reaction. If fused with sodium carbonate, the product mixture is leached with water. Insoluble carbonate separates from the water-soluble sodium sulfate.
Production Methods
Strontium carbonate, formed (1) by reaction of strontium salt solution and sodium carbonate or bicarbonate solution, (2) by reaction of strontium hydroxide solution and CO2. Strontium carbonate decomposes at 1,200 °C (2,192 °F) to form strontium oxide and CO2, and is dissolved by excess CO2, forming strontium bicarbonate, Sr(HCO3)2, solution.
Definition
strontianite: A mineral form ofstrontium carbonate, SrCO3.
General Description
Strontium carbonate is insoluble in water. It is used predominantly in producing other strontium salts.
General Description
Strontianite is a strontium carbonate mineral (SrCO3). It is the original and principal source of strontium. It often occurs in white masses of radiating fibres, although pale green, yellow, or gray colours are also known. Strontianite forms soft, brittle crystals that are commonly associated with barite, celestine, and calcite in low-temperature veins. Strontianite is used in pyrotechnics to impart a red colour and in sugar refining as a clarifying agent.
Strontium carbonate Preparation Products And Raw materials
Raw materials
1of3
chevron_rightPreparation Products
1of2
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| QUALIKEMS FINE CHEM PVT LTD | +911123618475 | New Delhi, India | 51 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| ShanPar Industries Pvt Ltd | +91-2652638973 +91-9909901206 | Gujarat, India | 115 | 58 | Inquiry |
| Merck Ltd | +91-2262109800 +91-2262109000 | Maharashtra, India | 272 | 58 | Inquiry |
| Divjyot Chemicals Private Limited | +91-8048603207 +91-9849191943 | Telangana, India | 11 | 58 | Inquiry |
| Kronox Lab Sciences Pvt Ltd | +91-9313231074 +91-9313231074 | Gujarat, India | 240 | 58 | Inquiry |
| Otto Chemie Pvt. Ltd. | +91 9820041841 | Mumbai, India | 5873 | 58 | Inquiry |
| Alfa Aesar | 1 800 209 7001 | Maharashtra, India | 6913 | 58 | Inquiry |
| Sisco Research Laboratories Pvt. Ltd. | +91-22-4268 5800 | Mumbai, India | 4317 | 58 | Inquiry |
| Central Drug House(P) Ltd. | 91-11-49404040 | New Delhi, India | 6160 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| QUALIKEMS FINE CHEM PVT LTD | 58 |
| JSK Chemicals | 58 |
| ShanPar Industries Pvt Ltd | 58 |
| Merck Ltd | 58 |
| Divjyot Chemicals Private Limited | 58 |
| Kronox Lab Sciences Pvt Ltd | 58 |
| Otto Chemie Pvt. Ltd. | 58 |
| Alfa Aesar | 58 |
| Sisco Research Laboratories Pvt. Ltd. | 58 |
| Central Drug House(P) Ltd. | 58 |
Related articles
- Strontium Carbonate: Applications in Bone Regeneration and its Biological Synthesis
- Fusarium oxysporum enables eco-friendly synthesis of strontium carbonate, vital in bioceramics for bone regeneration, showcasi....
- Jul 4,2024
1633-05-2(Strontium carbonate )Related Search:
1of4
chevron_right




