Lithium hydroxide monohydrate
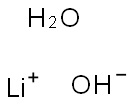
- CAS No.
- 1310-66-3
- Chemical Name:
- Lithium hydroxide monohydrate
- Synonyms
- Lithium hydroxide hydrate;ithium hydroxide monohydrate;Lithine hydrate;Lithium hydrate;Monohydrate Lithium Hydroxide;Li.HO.H2O;Lithium hydroxide mo;LITHIUM HYDROXIDE H2O;LITHIUM HYDROXIDE 1HYD;LITHIUM HYDROXIDE H2O ACS
- CBNumber:
- CB6260598
- Molecular Formula:
- Li.HO.H2O
- Molecular Weight:
- 41.96
- MOL File:
- 1310-66-3.mol
- Modify Date:
- 2024/6/21 13:10:12
| Melting point | 462 °C |
|---|---|
| Boiling point | 920 °C |
| Density | 1.51 |
| storage temp. | Store at room temperature. |
| solubility | H2O: 1 M at 20 °C, clear, colorless |
| form | Solid |
| color | White to cream or yellow |
| Specific Gravity | 1.51 |
| Odor | Odorless |
| PH Range | 14 |
| PH | ~12 (25℃, 1M in H2O) |
| Water Solubility | 109 g/L (20 ºC) |
| Sensitive | Air Sensitive & Hygroscopic |
| λmax |
λ: 260 nm Amax: 0.02 λ: 280 nm Amax: 0.02 |
| Merck | 14,5534 |
| Stability | Stable. Incompatible with moisture. strong acids, carbon dioxide. Absorbs carbon dioxide from the air. |
| InChIKey | WMFOQBRAJBCJND-UHFFFAOYSA-M |
| CAS DataBase Reference | 1310-66-3(CAS DataBase Reference) |
| NIST Chemistry Reference | LiOH(1310-66-3) |
| EPA Substance Registry System | Lithium hydroxide (Li(OH)), monohydrate (1310-66-3) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS05,GHS06 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H301-H314 | |||||||||
| Precautionary statements | P260-P270-P280-P303+P361+P353-P304+P340+P310-P305+P351+P338 | |||||||||
| Hazard Codes | C | |||||||||
| Risk Statements | 22-35-20/22-34-52 | |||||||||
| Safety Statements | 26-36/37/39-45-22-27 | |||||||||
| RIDADR | UN 2680 8/PG 2 | |||||||||
| WGK Germany | 2 | |||||||||
| RTECS | OJ6307080 | |||||||||
| F | 3-9-34 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 8 | |||||||||
| PackingGroup | II | |||||||||
| HS Code | 28252000 | |||||||||
| NFPA 704 |
|
Lithium hydroxide monohydrate price More Price(31)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | L4533 | Lithium hydroxide monohydrate BioXtra, 98.5-101.5% (titration) | 1310-66-3 | 100G | ₹5055.28 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | L4533 | Lithium hydroxide monohydrate BioXtra, 98.5-101.5% (titration) | 1310-66-3 | 500G | ₹8421.85 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | L4533 | Lithium hydroxide monohydrate BioXtra, 98.5-101.5% (titration) | 1310-66-3 | 1KG | ₹14418.9 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 62528 | Lithium hydroxide monohydrate BioUltra, ≥99.0% (T) | 1310-66-3 | 50G | ₹3669.68 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 62531 | Lithium hydroxide monohydrate puriss. p.a., ≥99.0% (T) | 1310-66-3 | 250G | ₹4730.53 | 2022-06-14 | Buy |
Lithium hydroxide monohydrate Chemical Properties,Uses,Production
Uses
Lithium hydroxide monohydrate is used for the production of lithium greases, lithium soaps, lithium stearate and lithium salts. It finds application as a carbon dioxide adsorbent in breathing gas purification systems for spacecrafts, submarines and rebreathers; as a storage-battery electrolyte; as a heat transfer medium and as a catalyst for polymerization reaction. It is also used in ceramics and some portland cement formulations.
Preparation
Lithium hydroxide monohydrate may also be made by the evaporation of a solution prepared by the reaction of calcium hydroxide and lithium carbonate.Anhydrous lithium hydroxide may be prepared by heating the monohydrate in air or in a vacuum.
General Description
Small colorless crystals. Denser than water. Contact may cause severe irritation to skin, eyes, and mucous membranes. Toxic by ingestion, inhalation and skin absorption. Used to make electric storage batteries, soaps, and lubricants.
An aqueous solution of a lithium salt below its boiling point has been considered for both a tritium breeding blanket and as a coolant in several fusion reactor designs. In order to breed sufficient tritium to fuel the reactor, a high concentration of lithium is desirable and lithium hydroxide is sufficiently soluble for this purpose. The solution would be contained in pipes and heat exchangers, most probably made from carbon steel.
Air & Water Reactions
Soluble in water; heat of dissolution may generate steam and cause spattering.
Reactivity Profile
LITHIUM HYDROXIDE MONOHYDRATE neutralizes acids exothermically to form salts plus water. Reacts with certain metals (such as aluminum and zinc) to form oxides or hydroxides of the metal and generate gaseous hydrogen. May initiate polymerization reactions in polymerizable organic compounds, especially epoxides. May generate flammable and/or toxic gases with ammonium salts, nitrides, halogenated organics, various metals, peroxides, and hydroperoxides. May serve as a catalyst. Reacts when heated above about 84°C with aqueous solutions of reducing sugars other than sucrose to evolve toxic levels of carbon monoxide [Bretherick, 5th Ed., 1995].
Health Hazard
TOXIC; inhalation, ingestion or skin contact with material may cause severe injury or death. Contact with molten substance may cause severe burns to skin and eyes. Avoid any skin contact. Effects of contact or inhalation may be delayed. Fire may produce irritating, corrosive and/or toxic gases. Runoff from fire control or dilution water may be corrosive and/or toxic and cause pollution.
Fire Hazard
Non-combustible, substance itself does not burn but may decompose upon heating to produce corrosive and/or toxic fumes. Some are oxidizers and may ignite combustibles (wood, paper, oil, clothing, etc.). Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated.
Purification Methods
It crystallises from hot water (3mL/g) as the monohydrate. It is dehydrated at 150o in a stream of CO2-free air. It sublimes at 220o with partial decomposition [Cohen Inorg Synth V 3 1957, Bravo Inorg Synth VII 1 1963].
Lithium hydroxide monohydrate Preparation Products And Raw materials
Raw materials
1of2
chevron_rightPreparation Products
1of4
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| GLR Innovations | +91 9891111994 | New Delhi, India | 4542 | 58 | Inquiry |
| PACIFIC ORGANICS PVT LTD | +91 9322467890 | Maharashtra, India | 51 | 58 | Inquiry |
| Vardhaman P Golechha | 9704422000 | Telangana, India | 294 | 58 | Inquiry |
| SNECOFRi Pvt Ltd | +91-9032850129 +91-9032850129 | Telangana, India | 404 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| Merck Ltd | +91-2262109800 +91-2262109000 | Maharashtra, India | 272 | 58 | Inquiry |
| Arvalli Castor Derivatives Pvt Ltd | +91-9898002809 +91-9898003829 | Vadodara, India | 9 | 58 | Inquiry |
| Rivashaa Agrotech Biopharma Pvt. Ltd. | +91-26463395 +91-7926462688 | Gujarat, India | 1615 | 58 | Inquiry |
| Guru Raghvendra Chemicals | +91-9849587687 +91-9849587687 | Hyderabad, India | 11 | 58 | Inquiry |
| Organo Metallics | +91-9391016448 +91-9705345345 | Telangana, India | 32 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| GLR Innovations | 58 |
| PACIFIC ORGANICS PVT LTD | 58 |
| Vardhaman P Golechha | 58 |
| SNECOFRi Pvt Ltd | 58 |
| JSK Chemicals | 58 |
| Merck Ltd | 58 |
| Arvalli Castor Derivatives Pvt Ltd | 58 |
| Rivashaa Agrotech Biopharma Pvt. Ltd. | 58 |
| Guru Raghvendra Chemicals | 58 |
| Organo Metallics | 58 |
Related articles
- Crystal structure of lithium hydroxide monohydrate
- Lithium hydroxide monohydrate, LiOH· H20, the "lithium hydrate" of commerce, was described crystallographically by V. von Lang....
- Apr 24,2024
- Property, preparation and application of lithium hydroxide monohydrate
- Lithium hydroxide monohydrate is one of the most important lithium compounds, is widely used in chemical raw materials, chemi....
- Jul 5,2022
1310-66-3(Lithium hydroxide monohydrate)Related Search:
1of4
chevron_right




