Potassium hydroxide
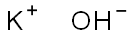
- CAS No.
- 1310-58-3
- Chemical Name:
- Potassium hydroxide
- Synonyms
- KOH;CAUSTIC POTASH;POTASH;POTTASIUM HYDROXIDE;Potassium hydroxide pellets;Potassio;Kaliumhydroxid;POTASIUM HYDROXIDE;Hydroxyde de potassium;KHO
- CBNumber:
- CB3107908
- Molecular Formula:
- KOH
- Molecular Weight:
- 56.11
- MOL File:
- 1310-58-3.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/6/7 16:47:56
| Melting point | 361 °C (lit.) |
|---|---|
| Boiling point | 1320°C |
| Density | 1.450 g/mL at 20 °C |
| vapor pressure | 1 mm Hg ( 719 °C) |
| refractive index |
n |
| Flash point | 52 °F |
| storage temp. | Store at +5°C to +30°C. |
| solubility | H2O: 1 M at 20 °C, clear, colorless |
| form | powder |
| Specific Gravity | 1.09 |
| color | white |
| Odor | Odorless |
| PH | 10.98(1 mM solution);11.95(10 mM solution);12.88(100 mM solution); |
| explosive limit | 3.5-15.0%(V) (ethanol) |
| Water Solubility | soluble |
| Sensitive | Air Sensitive & Hygroscopic |
| Merck | 14,7640 |
| Exposure limits | Ceiling in air 2 mg/m3 (ACGIH). |
| Dielectric constant | 3.3(Ambient) |
| Stability | Stable, but very hygroscopic. Dissolves exothermically in water. Incompatible with most metals, strong acids, acid chlorides, organic materials, zinc, aluminium, nitroalkanes, nitrobenzene, chlorine dioxide. Reacts vigorously with a wide variety of other materials. Readily absorbs water and carbon dioxide from the air. |
| InChIKey | KWYUFKZDYYNOTN-UHFFFAOYSA-M |
| LogP | -1.380 (est) |
| CAS DataBase Reference | 1310-58-3(CAS DataBase Reference) |
| NIST Chemistry Reference | Potassium hydroxide(1310-58-3) |
| EPA Substance Registry System | Potassium hydroxide (1310-58-3) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS05,GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H290-H302-H314 | |||||||||
| Precautionary statements | P234-P260-P280-P301+P312-P303+P361+P353-P305+P351+P338 | |||||||||
| Hazard Codes | C,F,T,Xi | |||||||||
| Risk Statements | 34-35-22-11-39/23/24/25-23/24/25-36/38-36/37-67-52/53 | |||||||||
| Safety Statements | 7-16-36/37-45-36/37/39-26-61 | |||||||||
| RIDADR | UN 2924 3/PG 2 | |||||||||
| OEL | Ceiling: 2 mg/m3 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | TT2100000 | |||||||||
| F | 3 | |||||||||
| Autoignition Temperature | 425 °C (ethanol) | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 8 | |||||||||
| PackingGroup | II | |||||||||
| HS Code | 28259090 | |||||||||
| Toxicity | LD50 orally in rats: 1.23 g/kg (Smyth) | |||||||||
| NFPA 704 |
|
Potassium hydroxide price More Price(106)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | P5958 | Potassium hydroxide BioXtra, ≥85% KOH basis | 1310-58-3 | 250G | ₹4698.05 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | P5958 | Potassium hydroxide BioXtra, ≥85% KOH basis | 1310-58-3 | 500G | ₹6679.03 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | P5958 | Potassium hydroxide BioXtra, ≥85% KOH basis | 1310-58-3 | 1KG | ₹8973.93 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | P1767 | Potassium hydroxide ≥85% KOH basis, pellets, white | 1310-58-3 | 500G | ₹2170 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | P1767 | Potassium hydroxide ≥85% KOH basis, pellets, white | 1310-58-3 | 250G | ₹3220 | 2022-06-14 | Buy |
Potassium hydroxide Chemical Properties,Uses,Production
Chemical Properties
Potassium hydroxide occurs as a white or nearly white fused mass. It is available in small pellets, flakes, sticks and other shapes or forms. It is hard and brittle and shows a crystalline fracture. Potassium hydroxide is hygroscopic and deliquescent; on exposure to air, it rapidly absorbs carbon dioxide and water with the formation of potassium carbonate. Soluble in water, alcohol, glycerol; slightly soluble in ether.
Indications
Potassium hydroxide (KOH) is a strong alkali that digests proteins and epidermal debris. In one study, 10% solution was applied b.i.d. to each lesion for 30 days with excellent clearance. The side effects included stinging of the lesion and one case of secondary infection. Also reported were the occurrence of a hypertrophic scar as well as some persistent or transitory hyper- and hypopigmentation. The same authors who used the 5% KOH solution completed further studies and they found it to be as effective-yet with decreased side effects.
Production Methods
Potassium hydroxide is made by the electrolysis of potassium chloride. Commercial grades may contain chlorides as well as other impurities.
Application
Potassium hydroxide is used as an emulsifier in lotions and as an alkali in liquid soaps, protective creams, and shaving preparations. Depending on the concentration used, it can be highly irritating to the skin and/or cause a burning sensation. It is used in making potassium salts, in electroplatingand lithography, in printing inks, as a mordantfor wood, and finds wide applicationsin organic syntheses and chemical analyses.
General Description
A white solid. Corrosive to metals and tissue. Used in soap manufacture, bleach, as an electrolyte in alkaline batteries, and as a food additive.
Air & Water Reactions
Hydrolysis generates enough heat to ignite adjacent combustible material [Haz. Chem. Data 1966]. Dissolves in water (with liberation of heat, may steam and spatter. Solution is basic (alkaline). Deliquescent
Reactivity Profile
POTASSIUM HYDROXIDE absorbs moisture readily forming caustic solution that attacks aluminum and zinc. A piece of potassium hydroxide causes liquid chlorine dioxide to explode [Mellor 2:289. 1946-47]. 1,2-dichloroethylene and potassium hydroxide forms chloroacetylene, which is explosive and spontaneously flammable in air. Potassium hydroxide is highly toxic [Rutledge 1968. p. 134]. A reaction between n-nitrosomethylurea and potassium hydroxide in n-butyl ether resulted in an explosion due to the formation of diazomethane [Schwab 1972]. Potassium persulfate and a little potassium hydroxide and water ignited a polythene (polyethylene) liner of a container by release of heat and oxygen [MCA Case History 1155. 1955]. Using potassium hydroxide to dry impure tetrahydrofuran, which contains peroxides, may be hazardous. Explosions have occurred in the past. Sodium hydroxide behaves in a similar way as potassium hydroxide [NSC Newsletter Chem. Soc. 1967]. A strong base. Forms caustic solution in water. [Merck 11th ed. 1989].
Health Hazard
Toxic by ingestion and inhalation, strong caustic, handle with gloves or tongs, corrosive to tissue. Eye, skin and upper respiratory tract irritant.
Potassium hydroxide is a strongly alkaline, hydrophilic substance and therefore solid potassium hydroxide is highly corrosive. It reacts with fat and can cause irreversible damage to any site of contact with the body (for example skin or eyes). Solutions of potassium hydroxide in water at concentrations above 0.5% (w/w) are irritating at points of contact and, at higher concentrations, the solutions can be corrosive. Potassium hydroxide does not cause skin allergies. Because of the corrosive properties of potassium hydroxide, its ingestion can be fatal. Under normal conditions of handling and use, potassium hydroxide in solution will dissociate into its constituent ions and, if ingested, will not be systemically available in the body as such.
Fire Hazard
Non-combustible, substance itself does not burn but may decompose upon heating to produce corrosive and/or toxic fumes. Some are oxidizers and may ignite combustibles (wood, paper, oil, clothing, etc.). Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated.
Flammability and Explosibility
Sodium hydroxide and potassium hydroxide are not flammable as solids or aqueous solutions.
Pharmaceutical Applications
Potassium hydroxide is widely used in pharmaceutical formulations
to adjust the pH of solutions. It can also be used to react with weak
acids to form salts.
Therapeutically, potassium hydroxide is used in various
dermatological applications.
Safety Profile
Poison by ingestion. An eye irritant and severe human skin irritant. Very corrosive to the eyes, skin, and mucous membranes. Mutation data reported. Ingestion may cause violent pain in throat and epigastrium, hematemesis, collapse. Stricture of esophagus may result if substance is not immedately fatal. Above 84' it reacts with reducing sugars to form poisonous carbon monoxide gas. Violent, exothermic reaction with water. Potentially explosive reaction with bromoform + crown ethers, chlorine dioxide, nitrobenzene, nitromethane, nitrogen trichloride, peroxidized tetrahydrofuran, 2,4,6- trinitrotoluene. Reaction with ammonium hexachloroplatinate(2-) + heat forms a heat- sensitive explosive product. Violent reaction or ignition under the appropriate condtions with acids, alcohols, p-bis(l,3- dbromoethyl)benzene, cyclopentadene, germanium, hyponitrous acid, maleic anhydride, nitroalkanes, 2-nitrophenol, potassium peroxodisulfate, sugars, 2,2,3,3- tetrafluoropropanol, thorium dicarbide. When heated to decomposition it emits toxic fumes of K2O. See also SODIUM HYDROXIDE.
Safety
Potassium hydroxide is widely used in the pharmaceutical and food
industries and is generally regarded as a nontoxic material at low
concentrations. At high concentrations it is a corrosive irritant to
the skin, eyes, and mucous membranes.
(rat, oral): 0.273 g/kg
Potential Exposure
KOH is generally used as an alkali and in the manufacture of other potassium compounds.
storage
splash goggles and impermeable gloves should be worn at all times when handling these substances to prevent eye and skin contact. Operations with metal hydroxide solutions that have the potential to create aerosols should be conducted in a fume hood to prevent exposure by inhalation. NaOH and KOH generate considerable heat when dissolved in water; when mixing with water, always add caustics slowly to the water and stir continuously. Never add water in limited quantities to solid hydroxides. Potassium hydroxide should be stored in an airtight, nonmetallic container in a cool, dry place, separated from acids and incompatible substances.
Shipping
UN1814 (solution) & UN1813 (solid); Potassium hydroxide, solid or solution, Hazard class: 8; Labels: 8-Corrosive material.
Purification Methods
Its carbonate content can be reduced by rinsing KOH sticks rapidly with water prior to dissolving them in boiled out distilled water. Alternatively, a slight excess of saturated BaCl2 or Ba(OH)2 can be added to the solution which, after shaking well, is set aside so that the BaCO3 is allowed to separate out. Davies and Nancollas [Nature 165 237 1950] rendered KOH solutions carbonate free by ion exchange using a column of Amberlite IR-100 in the OH-form.
Incompatibilities
Potassium hydroxide is a strong base and is incompatible with any compound that readily undergoes hydrolysis or oxidation. Violent reaction with acids, alcohols, water, metals (when wet), halogenated hydrocarbons; maleic anhydride. Heat is generated if KOH comes in contact with water and carbon dioxide from the air. It should not be stored in glass or aluminum containers, Corrosive to zinc, aluminum, tin and lead in the presence of moisture releasing combustible/explosive hydrogen gas. Can absorb water from air and give off sufficient heat to ignite surrounding combustible materials.
Waste Disposal
Dilute with large volume of water, neutralize and flush to sewer
Regulatory Status
GRAS listed. Accepted for use in Europe in certain food applications. Included in the FDA Inactive Ingredients Database (injections, infusions, and oral capsules and solutions). Included in nonparenteral and parenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Potassium hydroxide Preparation Products And Raw materials
Raw materials
1of3
chevron_rightPreparation Products
1of8
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Mumbai, India | 66 | 58 | Inquiry |
| ANJI BIOSCIENCES | +91-9000100077 +91-9000100077 | Hyderabad, India | 430 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| Kronox Lab Sciences Pvt Ltd | +91-9313231074 +91-9313231074 | Gujarat, India | 240 | 58 | Inquiry |
| Sree Rayalaseema HI Strength Hypo Ltd | +91-9848867692 +91-9848867692 | Andhra Pradesh, India | 19 | 58 | Inquiry |
| Annexe Chem Pvt Ltd | +91-9724624061 +91-9898722162 | Gujarat, India | 57 | 58 | Inquiry |
| HRV Global Life Sciences | +91-9820219686 +91-9820219686 | Telangana, India | 379 | 58 | Inquiry |
| Gajanan Enterprises | +91-8422892201 +91-8422892201 | Maharashtra, India | 8 | 58 | Inquiry |
| Sujata Chemicals | +91-9898025496 +91-9898025496 | Gujarat, India | 73 | 58 | Inquiry |
| Merck Ltd | +91-2262109800 +91-2262109000 | Maharashtra, India | 272 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| CLEARSYNTH LABS LTD. | 58 |
| ANJI BIOSCIENCES | 58 |
| JSK Chemicals | 58 |
| Kronox Lab Sciences Pvt Ltd | 58 |
| Sree Rayalaseema HI Strength Hypo Ltd | 58 |
| Annexe Chem Pvt Ltd | 58 |
| HRV Global Life Sciences | 58 |
| Gajanan Enterprises | 58 |
| Sujata Chemicals | 58 |
| Merck Ltd | 58 |
Related articles
- Introduction of Potassium hydroxide
- Potassium hydroxide is an inorganic compound which is denoted by the chemical formula KOH. Potassium hydroxide is also known a....
- Feb 28,2022
- Uses of Potassium hydroxide
- Potassium hydroxide is an inorganic compound with the formula KOH, and is commonly called caustic potash.
- Dec 24,2021
1310-58-3(Potassium hydroxide )Related Search:
1of4
chevron_right




