Ritalin
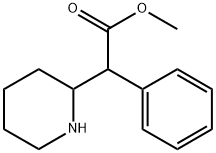
- CAS No.
- 113-45-1
- Chemical Name:
- Ritalin
- Synonyms
- DEXMETHYLPHENIDATE;Rubifen;Phenidylate;Methylphenidan;methyl 2-phenyl-2-(piperidin-2-yl)acetate;alpha-Phenyl-2-piperidineacetic acid methyl ester;C07196;meridil;Calocain;4311/b Ciba
- CBNumber:
- CB1874770
- Molecular Formula:
- C14H19NO2
- Molecular Weight:
- 233.31
- MOL File:
- 113-45-1.mol
- Modify Date:
- 2023/5/4 17:34:31
| Melting point | 74.5°C |
|---|---|
| Boiling point | 327℃ |
| Density | 1.070 |
| refractive index | 1.5110 (estimate) |
| Flash point | 152℃ |
| pka | 9.51±0.10(Predicted) |
| CAS DataBase Reference | 113-45-1 |
| EPA Substance Registry System | 2-Piperidineacetic acid, ?-phenyl-, methyl ester (113-45-1) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS08 |
|---|---|
| Signal word | Warning |
| Hazard statements | H315-H302-H319-H341 |
| Precautionary statements | P264-P280-P305+P351+P338-P337+P313P-P201-P202-P281-P308+P313-P405-P501-P264-P280-P302+P352-P321-P332+P313-P362-P264-P270-P301+P312-P330-P501 |
| Toxicity | LD50 oral in rat: 367mg/kg |
Ritalin Chemical Properties,Uses,Production
History
Methylphenidate is a piperidine derivative that is a central nervous system stimulant. Methylphenidate, in its hydrochloride form, is the active ingredient in the common medication Ritalin. Methylphenidate was first synthesized from benzyl cyanide and 2-chloropyridine in Basel, Switzerland in 1944 by Ciba chemist Leandro Panizzon (1907 ?).
Panizzon named the substance Ritaline after his wife, whose nickname was Rita. Panizzon and Max Hartmann proposed an improved synthesis for methylphenidate and obtained a U.S. patent for its preparation in 1950 (U.S. Patent Number 2507631). In 1954, methylphenidate was patented for use as an agent for treating psychological disorders under the name Ritalin by the Ciba pharmaceutical company.(Ciba subsequently merged with the Geigy company to become Ciba-Geigy, and then Ciba-Geigy merged with Sandoz to form Novartis.) Methylphenidate was first used to reverse drug-induced coma. Ritalin was approved by the Food and Drug Administration (FDA) in 1955 and introduced in the United States in 1956 for several conditions including depression, senile behavior, lethargy, and narcolepsy.
Hyperactive children were first treated with stimulants in 1937. Since methylphenidate first appeared on the market, its use for treating ADHD has steadily increased. It is the most commonly used medication for treating children diagnosed with ADHD.
Uses
Stimulant.
World Health Organization (WHO)
Methylphenidate, a piperidine derivative with mild central stimulant activity, was introduced in 1956. Its pharmacological properties resemble those of amfetamines and it shares their abuse potential. Methylphenidate retains a place as an adjunct in the treatment of hyperkinetic syndromes in both children and adults. It is controlled under Schedule II of the 1971 Convention on Psychotropic Substances. (Reference: (UNCPS2) United Nations Convention on Psychotropic Substances (II), , , 1971)
Safety Profile
Poison experimentally by ingestion, intraperitoneal, intravenous, and subcutaneous routes. Moderately toxic to humans by intravenous route. Human systemic effects by intravenous route: dyspnea. An experimental teratogen. When heated to decomposition it emits toxic fumes of NOx.
Ritalin Preparation Products And Raw materials
Raw materials
1of2
chevron_right




