Afatinib dimaleate
- CAS No.
- 850140-73-7
- Chemical Name:
- Afatinib dimaleate
- Synonyms
- Alfatinib maleate;Afatinib (diMaleate);BAY-2416964;BIBW2992-MA2;Afatinib DimaL;Aftinib maleate;Afatinib DIMELATE;BIBW2992 DiMaleate;Afatinib (maleate);BIBW2993 DiMaleate
- CBNumber:
- CB22630067
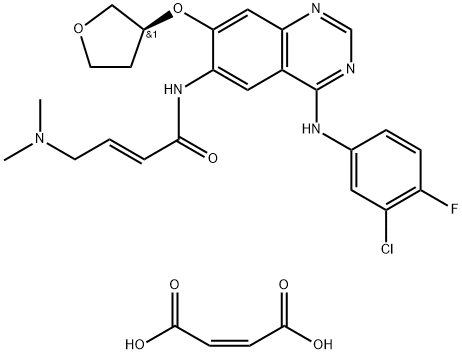
- Molecular Formula:
- C28H29ClFN5O7
- Molecular Weight:
- 602.02
- MOL File:
- 850140-73-7.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/9/22 21:01:39
| Melting point | >237oC (dec.) |
|---|---|
| storage temp. | Refrigerator |
| solubility | DMSO (Slightly), Methanol (Slightly) |
| form | Solid |
| color | White to Pale Yellow |
| InChI | InChI=1/C24H25ClFN5O3.C4H4O4/c1-31(2)8-3-4-23(32)30-21-11-17-20(12-22(21)34-16-7-9-33-13-16)27-14-28-24(17)29-15-5-6-19(26)18(25)10-15;5-3(6)1-2-4(7)8/h3-6,10-12,14,16H,7-9,13H2,1-2H3,(H,30,32)(H,27,28,29);1-2H,(H,5,6)(H,7,8)/b4-3+;2-1-/t16-;/s3 |
| InChIKey | LIENDGDDWJRJLO-LBXKZPGENA-N |
| SMILES | C(/C(=O)O)=C/C(=O)O.N(C1C=CC(F)=C(Cl)C=1)C1=NC=NC2C=C(O[C@@H]3COCC3)C(NC(=O)/C=C/CN(C)C)=CC1=2 |&1:25,r| |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS07,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H362-H319-H302-H372-H361 | |||||||||
| Precautionary statements | P201-P260-P263-P264-P270-P308+P313-P264-P280-P305+P351+P338-P337+P313P-P264-P270-P301+P312-P330-P501-P260-P264-P270-P314-P501-P201-P202-P281-P308+P313-P405-P501 | |||||||||
| HS Code | 29339900 | |||||||||
| NFPA 704 |
|
Afatinib dimaleate Chemical Properties,Uses,Production
Description
Afatinib dimaleate was approved by the U.S. Food and Drug Administration (FDA) in 2013 for the treatment of non-small cell lung cancer (NSCLC). Specifically, it was approved for patients presenting with metastatic NSCLC tumors which contain epidermal growth factor receptor (EGFR) exon deletions or exon 21 mutations. Afatinib dimaleate is a covalent inhibitor of ErbB tyrosine kinases (tyk), which downregulates ErbB signaling by irreversible binding of EGFR tyk binding sites. While no manufacturing route has been disclosed to date, the most scalable published route likely derives from two Boehringer Ingelheim patents.
Uses
Afatinib Dimaleate is a salt of Afatinib {BIBW 2992), an aminocrotonylamino-substituted quinazoline derivative used for treating cancer and diseases of the respiratory tract, lungs, gastrointestinal tract, bile duct, and gallbladder. An anilino-quinazoline that irreversibly inhibits EGFR and HER2 kinase activity.
Definition
ChEBI: Afatinib dimaleate is a maleate salt obtained by combining afatinib with two molar equivalents of maleic acid. Used for the first-line treatment of patients with metastatic non-small cell lung cancer. It has a role as a tyrosine kinase inhibitor and an antineoplastic agent. It contains an afatinib.
General Description
Afatinib dimaleate is the dimaleate form of afatinib. It is a tyrosine kinase inhibitor of epidermal growth factor receptors (ERBB RECEPTORS) and an anti-angiogenic agent. Afatinib dimaleate has anti-tumour activity and is mainly used for the treatment of patients with metastatic non-small cell lung cancer (which has metastasised or spread to other parts of the body), or certain tumours with mutations in the EGFR gene and patients whose disease has worsened after platinum-based chemotherapy. It is also used for the treatment of esophageal squamous cell carcinoma (ESCC) and gastric cancer.
Biological Activity
Afatinib dimaleate is an orally bioavailable and irreversible dual-specific inhibitor of the ErbB family (EGFR and HER2). IC50 are 0.4, 0.5, 10, 14, 1 nM for EGFRL858R, EGFRwt, EGFR L858R/T790M, ErbB2 (HER2) and ErbB4 (HER4), respectively. Afatinib dimaleate is available for cancer (esophageal squamous cell carcinoma (ESCC), non-small cell lung cancer (NSCLC) and gastric cancer) studies.
Side effects
Afatinib dimaleate is similar to afatinib in terms of side effects. Common side effects include: diarrhoea, fever, difficulty urinating, burning or pain, redness, swelling or pain in the skin, discharge or excessive tearing, burning, dryness or itching of the eyes, and skin ulcers. Occasional symptoms of coughing and difficulty breathing. A very small number of patients may have bloody stools, black stools or tarry stools, indigestion, nausea, severe abdominal pain, cramping or burning sensation, and vomiting of a substance that looks like coffee grounds. Please contact your doctor promptly if any serious adverse reactions occur.
Afatinib dimaleate Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Shilpa Medicare Limited (SML) | +91-8532238704 +91-9320649838 | Karnataka, India | 61 | 58 | Inquiry |
| Beta Drugs | +91-9316590666 +91-7015991921 | Haryana, India | 34 | 58 | Inquiry |
| SynZeal Research Pvt Ltd | +1 226-802-2078 | Gujarat, India | 6522 | 58 | Inquiry |
| Pharmaffiliates Analytics and Synthetics P. Ltd | +91-172-5066494 | Haryana, India | 6773 | 58 | Inquiry |
| Triveni Interchem Private Limited (Group Of Triveni... | 08046042087 | Gujarat, India | 497 | 58 | Inquiry |
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| A.J Chemicals | 91-9810153283 | New Delhi, India | 6124 | 58 | Inquiry |
| Pharma Affiliates | 172-5066494 | Haryana, India | 6761 | 58 | Inquiry |
| HETERO LABS LTD | (630) 931-6800 | New Delhi, India | 97 | 58 | Inquiry |
| FRESENIUS KABI ONCOLOGY LTD | +86-571-28971376 | New Delhi, India | 21 | 58 | Inquiry |
Related articles
- Afatinib dimaleate: Chemical properties, Indications, Mechanism of action and Side effects
- Afatinib dimaleate is very soluble in water and aqueous buffers with a pH of 1 to 6 (> 50 mg/ mL). The free base is strongly l....
- Sep 2,2024
- BIBW2992 DiMaleate: pharmacokinetic, clinical applications and safety
- BIBW2992 DiMaleate is a well-tolerated medication for certain types of lung cancer, showing bioequivalence and efficacy in cli....
- Nov 20,2023
- BIBW2992 Dimaleate - Pharmacodynamics
- Afatinib dimaleate (Tovok; BIBW2992; Gilotrif) is a salt form of Afatinib. Afatinib is a second-generation, orally administere....
- Dec 13,2019
850140-73-7(Afatinib dimaleate)Related Search:
1of4
chevron_right




