Barium acetate
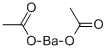
- CAS No.
- 543-80-6
- Chemical Name:
- Barium acetate
- Synonyms
- octanbarnaty;BARIUM ACETATE;BariumAcetateGr;Barium diacetate;BariumAcetateA.R.;Bariumacetate,99%;BARIUM ACETATE 99%;BARIUM ACETATE XTL;BARIUM ACETATE, ACS;BariumAcetateAcs/Ar
- CBNumber:
- CB2450535
- Molecular Formula:
- C4H6BaO4
- Molecular Weight:
- 255.42
- MOL File:
- 543-80-6.mol
- MSDS File:
- SDS
- Modify Date:
- 2023/8/31 16:33:09
| Melting point | 450°C |
|---|---|
| Density | 2.46 |
| vapor pressure | 15.7hPa at 25℃ |
| storage temp. | Store at +5°C to +30°C. |
| solubility | 720g/l |
| form | Solid |
| Specific Gravity | 2.468 |
| color | White |
| PH | 7-8.5 (20℃, 5%) |
| Odor | Slight acetic acid odor |
| PH Range | 7 - 8.5 at 50 g/l at 25 °C |
| Water Solubility | soluble |
| Merck | 14,965 |
| BRN | 3693411 |
| Exposure limits |
ACGIH: TWA 0.5 mg/m3 NIOSH: IDLH 50 mg/m3; TWA 0.5 mg/m3 |
| Stability | Stable. Incompatible with strong oxidizing agents, acids. |
| InChIKey | ITHZDDVSAWDQPZ-UHFFFAOYSA-L |
| LogP | -0.17 |
| CAS DataBase Reference | 543-80-6(CAS DataBase Reference) |
| EPA Substance Registry System | Barium acetate (543-80-6) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H302+H332 | |||||||||
| Precautionary statements | P261-P264-P270-P271-P301+P312-P304+P340+P312 | |||||||||
| Hazard Codes | Xn | |||||||||
| Risk Statements | 20/22 | |||||||||
| Safety Statements | 28-28A | |||||||||
| RIDADR | 1564 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | AF4550000 | |||||||||
| TSCA | No | |||||||||
| HS Code | 2915 29 00 | |||||||||
| HazardClass | 6.1 | |||||||||
| PackingGroup | III | |||||||||
| Toxicity | LD50 orally in Rabbit: 921 mg/kg | |||||||||
| NFPA 704 |
|
Barium acetate price More Price(15)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 255912 | Barium acetate 99.999% trace metals basis | 543-80-6 | 10G | ₹8605.88 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 255912 | Barium acetate 99.999% trace metals basis | 543-80-6 | 50G | ₹32063.65 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 243671 | Barium acetate ACS reagent, 99% | 543-80-6 | 100G | ₹4600.63 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 243671 | Barium acetate ACS reagent, 99% | 543-80-6 | 500G | ₹9634.25 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 1.01704 | Barium acetate for analysis EMSURE? ACS | 543-80-6 | 500G | ₹14790 | 2022-06-14 | Buy |
Barium acetate Chemical Properties,Uses,Production
Description
Barium acetate (Ba(C2H3O2)2) is the salt of barium (II) and acetic acid.
Chemical Properties
Barium acetate is a white powder, which is highly soluble: at 0 °C, 55.8 g of barium acetate can be dissolved in 100 g of water. It decomposes upon heating into barium carbonate .
Physical properties
White powdery solid; density 2.47g/cm3; decomposes on heating; highly soluble in water (55.8g /100g at 0°C), sparingly soluble in methanol (~1.43 g per liter).
Uses
Barium acetate is used as a mordant for printing textile fabrics, for drying paints and varnishes and in lubricating oil. In chemistry, it is used in the preparation of other acetates; and as a catalyst in organic synthesis.
Preparation
Barium acetate is generally produced by the reaction of acetic acid with barium carbonate :
BaCO3 + 2 CH3COOH → (CH3COO)2Ba + CO2 + H2O
The reaction is performed in solution and the barium acetate crystallizes out. Alternatively, barium sulfide can be used :
BaS + 2 CH3COOH → (CH3COO)2Ba +H2S
Again, the solvent is evaporated off and the barium acetate crystallized.
Reactions
When heated in air, barium acetate decomposes to the carbonate. It reacts with acids: reaction with sulfuric acid, hydrochloric acid and nitric acid give the sulfate, chloride and nitrate respectively.
General Description
Barium acetate is the barium salt of acetic acid.
Hazard
The salt or its aqueous solution is highly toxic. LD10 (oral) rabbit: 236 mg/kg; LD10 (subcutaneous) rabbit: 96 mg/kg. See Barium.
Safety Profile
Poison via ingestion, intravenous, and subcutaneous routes. When heated to decomposition it emits acrid smoke and fumes. See also BARIUM COMPOUNDS.
Purification Methods
Crystallise the salt twice from anhydrous acetic acid and dry it under vacuum for 24hours at 100o. [Beilstein 2 I 49, 2 II 117, 2 III 192, 2 IV 114.]
Barium acetate Preparation Products And Raw materials
Raw materials
Preparation Products
1of2
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| GLR Innovations | +91 9891111994 | New Delhi, India | 4542 | 58 | Inquiry |
| ALLIPO CHEMICALS | +91-8238998187 +91-8238998187 | Gujarat, India | 32 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| Kronox Lab Sciences Pvt Ltd | +91-9313231074 +91-9313231074 | Gujarat, India | 240 | 58 | Inquiry |
| New Alliance Dye Chem Pvt Ltd | +91-9820112338 +91-9820112338 | Maharashtra, India | 104 | 58 | Inquiry |
| Nithyasri Chemicals (APURVA CHEMICALS) | +91-2512692130 +91-9323652199 | Maharashtra, India | 75 | 58 | Inquiry |
| A.B.ENTERPRISES | +91-9223381464 +91-9223381464 | Maharashtra, India | 78 | 58 | Inquiry |
| Divjyot Chemicals Private Limited | +91-8048603207 +91-9849191943 | Telangana, India | 11 | 58 | Inquiry |
| RYZE CHEMIE | +91-9702966440 +91-9702966440 | Maharashtra, India | 335 | 58 | Inquiry |
| FINAR LTD | +91 - 2717 - 616717 | New Delhi, India | 660 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| GLR Innovations | 58 |
| ALLIPO CHEMICALS | 58 |
| JSK Chemicals | 58 |
| Kronox Lab Sciences Pvt Ltd | 58 |
| New Alliance Dye Chem Pvt Ltd | 58 |
| Nithyasri Chemicals (APURVA CHEMICALS) | 58 |
| A.B.ENTERPRISES | 58 |
| Divjyot Chemicals Private Limited | 58 |
| RYZE CHEMIE | 58 |
| FINAR LTD | 58 |
543-80-6(Barium acetate)Related Search:
1of4
chevron_right




