Ammonium acetate
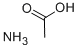
- CAS No.
- 631-61-8
- Chemical Name:
- Ammonium acetate
- Synonyms
- NH4OAC;Ammonium ethanoate;Ammoniumacetat;Ammonium aletate;acetated’ammonium;NH Ac;MARKER DNA 1KBP;AMMONIUM ACETATE;ACS,Reag. Ph Eur;AmmoniumAcetateGr
- CBNumber:
- CB8438173
- Molecular Formula:
- C2H7NO2
- Molecular Weight:
- 77.08
- MOL File:
- 631-61-8.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/6/21 13:10:12
| Melting point | 110-112 °C (dec.) (lit.) |
|---|---|
| Boiling point | 138.46°C (rough estimate) |
| Density | 1.07 g/mL at 20 °C |
| vapor pressure | 0.017-0.02Pa at 25℃ |
| refractive index | 1.4350 (estimate) |
| Flash point | 136 °C |
| storage temp. | Store at +15°C to +25°C. |
| solubility | H2O: 1 M at 20 °C, clear, colorless |
| form | Solid |
| pka | 4.6(Acetic Acid), 9.3(Ammonium Hydroxide)(at 25℃) |
| color | White |
| PH Range | 6.7 - 7.3 |
| Odor | Slight acetic acid odor |
| PH | 7(1 mM solution);7(10 mM solution);7(100 mM solution);6.95(1000 mM solution) |
| Water Solubility | 1480 g/L (20 ºC) |
| Sensitive | Hygroscopic |
| λmax |
λ: 260 nm Amax: 0.015 λ: 280 nm Amax: 0.01 |
| Merck | 14,495 |
| BRN | 4186741 |
| Stability | Hygroscopic |
| InChIKey | USFZMSVCRYTOJT-UHFFFAOYSA-N |
| LogP | -2.79 |
| CAS DataBase Reference | 631-61-8(CAS DataBase Reference) |
| NIST Chemistry Reference | Ammonium acetate, anhydrous(631-61-8) |
| EPA Substance Registry System | Ammonium acetate (631-61-8) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H319 | |||||||||
| Precautionary statements | P305+P351+P338 | |||||||||
| Safety Statements | 24/25 | |||||||||
| RIDADR | UN 9079 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | AF3675000 | |||||||||
| F | 3 | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 29152900 | |||||||||
| Toxicity | LD i.v. in mice: 1.8 mg (NH4+)/20g (Welch) | |||||||||
| NFPA 704 |
|
Ammonium acetate price More Price(82)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | A7330 | Ammonium acetate BioXtra, ≥98% | 631-61-8 | 100G | ₹2024.28 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | A7330 | Ammonium acetate BioXtra, ≥98% | 631-61-8 | 500G | ₹4535.68 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | A7330 | Ammonium acetate BioXtra, ≥98% | 631-61-8 | 2.5KG | ₹12188.95 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | A7262 | Ammonium acetate reagent grade, ≥98% | 631-61-8 | 500G | ₹6852.23 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | A7262 | Ammonium acetate reagent grade, ≥98% | 631-61-8 | 5KG | ₹20599.98 | 2022-06-14 | Buy |
Ammonium acetate Chemical Properties,Uses,Production
Chemical Properties
Ammonium acetate is a white, deliquescent crystalline solid with a slightly acetic odor. It is used as a reagent in analytical chemistry for determining lead and iron content,a chemical intermediate in manufacturing acetamide. It is also a diuretic in veterinary medicine, in other drugs, in textile dyeing, in meat preservative, in foam rubbers, in vinyl plastics, in stripping explosives, and in determining lead and iron content.

Physical properties
White crystalline solid; deliquescent; melts at 114°C; decomposes at elevated temperatures; density 1.17 g/cm3 at 20°C, density of a 10% solution 1.022 g/mL, and a 50% solution 1.092 g/mL; very soluble in cold water (1,480 g/L at 4°C); also soluble in cold alcohol and acetone (78.9 g/L in methanol at 15°C); solution loses ammonia on standing and becomes acidic.
Uses
Ammonium acetate is used in the manufacture of acetamide and as a diuretic and diaphoretic in medical applications. The wool industry also uses this salt as a dye mordant.
Buffer solution; determination of lead and iron; separating lead sulfate from other sulfates.
Preparation
Ammonium acetate is manufactured by neutralizing acetic acid with ammonium carbonate or by passing ammonia gas into glacial acetic acid. Acidic ammonium acetate, CH3CO2NH4.CH3CO2H[25007-86-7], is manufactured by dissolving the neutral salt in acetic acid.
General Description
A white crystalline solid. The primary hazard is the threat to the environment. Immediate steps should be taken to limit its spread to the environment. Ammonium acetate is used in chemical analysis, in pharmaceuticals, in preserving foods, and for other uses.
Air & Water Reactions
Water soluble.
Reactivity Profile
Ammonium acetate causes the decomposition of sodium hypochlorite within a few seconds [Mellor 2 Supp. 1:550 1956].
Health Hazard
Inhalation of dust irritates nose and mouth. Ingestion irritates mouth and stomach. Contact with dust causes irritation of eyes and mild irritation of skin.
Fire Hazard
Special Hazards of Combustion Products: Irritating vapors of ammonia acetic acid, and nitrogen oxides may form in fires.
Safety Profile
Poison by intravenous route. Moderately toxic by intraperitoneal route. When heated to decomposition it emits toxic fumes of NO, and NH3
Potential Exposure
Ammonium acetate is used as a chemical reagent, to make drugs; foam rubber; vinyl plastics; explosives, and to preserve foods. An environmental threat.
Shipping
UN3077 Environmentally hazardous substances, solid, n.o.s., Hazard class: 9; Labels: 9-Miscellaneous hazardous material, Technical Name Required.
Purification Methods
Crystallise it twice from anhydrous acetic acid, and dry under vacuum for 24hours at 100o [Proll & Sutcliff Trans Faraday Soc 57 1078 1961].
Incompatibilities
Combustible solid. Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, epoxides, sodium hypochlorite, potassium chlorate, sodium nitrite.
Ammonium acetate Preparation Products And Raw materials
Raw materials
Preparation Products
1of8
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| GLR Innovations | +91 9891111994 | New Delhi, India | 4542 | 58 | Inquiry |
| SAGAR LIFE SCIENCES PVT LTD | +91-8770359041 +91-8770359041 | Gujarat, India | 79 | 58 | Inquiry |
| ADVENT CHEMBIO PRIVATE LIMITED | +91-9821524116 +91-9821524116 | Mumbai, India | 353 | 58 | Inquiry |
| Yogi Dye Chem Industries | +91-9821098577 +91-9821098577 | Mumbai, India | 124 | 58 | Inquiry |
| PERFECT CHEMICAL | +91-9820465461 +91-9820465461 | Mumbai, India | 391 | 58 | Inquiry |
| ALLIPO CHEMICALS | +91-8238998187 +91-8238998187 | Gujarat, India | 32 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| AVA CHEMICALS PVT LTD | +91-8879390046 +91-8879390046 | Maharashtra, India | 74 | 58 | Inquiry |
| New Alliance Dye Chem Pvt Ltd | +91-9820112338 +91-9820112338 | Maharashtra, India | 104 | 58 | Inquiry |
| Sampan Enterprises | +91-9821234951 +91-9833144093 | Maharashtra, India | 6 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| GLR Innovations | 58 |
| SAGAR LIFE SCIENCES PVT LTD | 58 |
| ADVENT CHEMBIO PRIVATE LIMITED | 58 |
| Yogi Dye Chem Industries | 58 |
| PERFECT CHEMICAL | 58 |
| ALLIPO CHEMICALS | 58 |
| JSK Chemicals | 58 |
| AVA CHEMICALS PVT LTD | 58 |
| New Alliance Dye Chem Pvt Ltd | 58 |
| Sampan Enterprises | 58 |
Related articles
- Learn More About Ammonium Acetate
- The passage introduces the physical and chemical properties, uses and hazards of Ammonium acetate.
- Nov 22,2022
- How long does 10M ammonium acetate take to dissolve in water?
- I need to make 10M ammonium acetate for DNA extraction. I calculated the amount required for 70 ml solution and started dissol....
- Aug 30,2019
Related Qustion
- Q:Is Ammonium acetate an acid or base and what is the value of pH?
- A:Ammonium acetate is a salt that is a combination of a weak acid and a weak base. When dissolved in water, ammonium acetate has....
- Apr 28,2024
631-61-8(Ammonium acetate)Related Search:
1of4
chevron_right




