Chlorhexidine
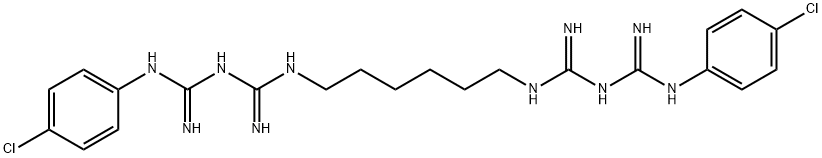
- CAS No.
- 55-56-1
- Chemical Name:
- Chlorhexidine
- Synonyms
- CHLORHEXIDINE BASE;CHLOROHEXIDINE;chlorhexidin;sterilon;fimeil;soretol;hexadol;nolvasan;tubulicid;rotersept
- CBNumber:
- CB4732251
- Molecular Formula:
- C22H30Cl2N10
- Molecular Weight:
- 505.45
- MOL File:
- 55-56-1.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/7/26 15:15:06
| Melting point | 134-136 °C (lit.) |
|---|---|
| Boiling point | 641.45°C (rough estimate) |
| Density | 1.1555 (rough estimate) |
| refractive index | 1.6300 (estimate) |
| storage temp. | 2-8°C |
| solubility | water: soluble0.08% at 20°C |
| form | Solid |
| pka | pKa 10.78 (Uncertain) |
| color | White to off-white |
| Water Solubility | 0.08 g/100 mL (20 ºC) |
| Merck | 13,2108 |
| BRN | 2826432 |
| Stability | Stable. Incompatible with strong oxidizing agents. |
| InChIKey | GHXZTYHSJHQHIJ-UHFFFAOYSA-N |
| LogP | 0.080 |
| CAS DataBase Reference | 55-56-1(CAS DataBase Reference) |
| EPA Substance Registry System | Chlorhexidine (55-56-1) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS05,GHS09 |
|---|---|
| Signal word | Danger |
| Hazard statements | H318-H410 |
| Precautionary statements | P273-P280-P305+P351+P338-P391-P501 |
| Hazard Codes | Xi,N |
| Risk Statements | 36/37/38-43-51/53-50/53 |
| Safety Statements | 26-36/37-60-61 |
| RIDADR | UN 3077 9/PG 3 |
| WGK Germany | 3 |
| RTECS | DU1925000 |
| F | 10-34 |
| HazardClass | 9 |
| PackingGroup | III |
| HS Code | 29215900 |
| Toxicity | LD50 orally in Rabbit: 5000 mg/kg |
Chlorhexidine price More Price(5)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 8.43969 | Chlorohexidine for synthesis | 55-56-1 | 50G | ₹6290 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | PHR1421 | Chlorhexidine Pharmaceutical Secondary Standard; Certified Reference Material | 55-56-1 | 1G | ₹8281.13 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 8.43969 | Chlorohexidine for synthesis | 55-56-1 | 250G | ₹20450 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 282227 | Chlorhexidine ≥99.5% | 55-56-1 | 1G | ₹12925.05 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 282227 | Chlorhexidine ≥99.5% | 55-56-1 | 5G | ₹33633.28 | 2022-06-14 | Buy |
Chlorhexidine Chemical Properties,Uses,Production
Description
Chlorhexidine is a cationic broad-spectrum antimicrobial agent belonging to the bis(biguanide) family. Its mechanism of action involves destabilization of the outer bacterial membrane. It is effective on both Gram-positive and Gram-negative bacteria, although it is less effective with some Gram-negative bacteria. It has both bactericidal and bacteriostatic mechanisms of action.
Chlorhexidine's antimicrobial effects are associated with the attractions between chlorhexidine (cation) and negatively charged bacterial cells. After chlorhexidine is absorpted onto the organism's cell wall, it disrupts the integrity of the cell membrane and causes the leakage of intracellular components of the organisms.
Aqueous solutions of chlorhexidine are most stable within the pH range of 5-8. Above pH 8.0 chlorhexidine base is precipitated and in more acid conditions there is gradual deterioration of activity because the compound is less stable. Chlorhexidine is used primarily as a topical antiseptic/disinfectant in wound healing, at catheterization sites, in various dental applications and in surgical scrubs. It has a LD-50 orally in mice as a diacetate at 2gm./kg. In digluconate form the LD-50 is 1800 gm./kg.
Chemical Properties
Chlorhexidine occurs as an odorless, bitter tasting, white crystalline powder.
Chemical Properties
Chlorhexidine [55-56-1], 1,6-bis(4-chlorophenylbiguanido)hexane, C22H30Cl2N10, Mr 505.45, mp 134 ℃, is a widely used antiseptic (→Disinfectants). It can be prepared, for example, from 1,6-hexamethylenebis(dicyandiamide) and 4-chloroaniline hydrochloride. Chlorhexidine is used primarily as its salts (e.g., the dihydrochloride, diacetate, and digluconate) in disinfectants (disinfection of the skin and hands), cosmetics (additive to creams, toothpaste, deodorants, and antiperspirants), and pharmaceutical products (preservative in eyedrops, active substance in wound dressings and antiseptic mouthwashes).
Uses
Chlorhexidine is an antibacterial used for numerous applications. It is a cationic polybiguanide (bisbiguanide) used primarily as its salts, dihydrochloride, diacetate, and digluconate. Chlorhexidine is one of those drugs which are enlisted/included in the World Health Organization's List of Essential Medicines, a list of the most important drugs needed in a basic health system.
- Chlorhexidine is used as a germicidal compound in teat dips. Also used as navel treatment, udder and eye wash, surgical scrub and sterilization material.
- Chlorhexidine is used primarily as a topical antiseptic/disinfectant in wound healing, at catheterization sites, in various dental applications and in surgical scrubs. it is used as an antibacterial agent in humans to control gingivitis and over all plaque control in preventative dentistry.
- Hydrogenolysis of benzyl-nitrogen bonds.
- Bacteriostatic;Detergent.
Indications
This topical antiseptic product acts rapidly but, like hexachlorophene, persists on the skin to give a cumulative, continuing antibacterial effect. Like iodophors and alcohol, it is active against gram-positive and gram-negative bacteria, including P. aeruginosa, as well as common yeasts and fungi. It does not lose effectiveness in the presence of whole blood. Many consider it the antiseptic of choice for skin cleansing and surgical scrubs. Contact allergy is not uncommon. Chlorhexidine should not be used near the eyes or mucosal surfaces, because it may cause irritation or even anaphylaxis.
Production Methods
Chlorhexidine may be prepared either by condensation of polymethylene bisdicyandiamide with 4-chloroaniline hydrochloride or by condensation of 4-chlorophenyl dicyandiamine with hexamethylenediamine dihydrochloride. Chlorhexidine may also be synthesized from a series of biguanides.
Definition
ChEBI: A bisbiguanide compound with a structure consisting of two (p-chlorophenyl)guanide units linked by a hexamethylene bridge.
Application
Chlorhexidine is an important medical, dental and pharmaceutical antiseptic, disinfectant and preservative. It is bactericida and fungicidalsy but does not kill bacterial spores or mycobacteria, although it inhibits growth. It has a low order of activity against viruses, but high concentrations are effective in killing cysts of Acanthamoeba spp., organisms of potential clinical significance to the wearers of contact lenses. Properties Chlorhexidine is a bisbiguanide which is available as the acetate (diacetate), hydrochloride and gluconate salts. These are stable in solution and can be autoclaved although small amounts of chloroaniline are released.” As a cationic agent, chlorhexidine is incompatible with anionic surfactants and its antimicrobial activity is reduced in the presence of non-ionic surface-active agents. Activity is also reduced or abolished by phospholipids (a factor of significance in neutralizing chlorhexidine activity during the performance of biocidal tests) and by organic matter including serum. Some of these aspects have been well documented in the recent comprehensive paper of Nicoletti et al.” They also point out that the efficacy of chlorhexidine is influenced by formulation components and by the composition of the culture medium in which minimum inhibitory concentrations (MICs) are determined.
General Description
Effect of the chlorhexidine:hydroxypropyl-β-cyclodextrin inclusion compound on in vitro slabs of bovine dentine has been investigated.
Pharmaceutical Applications
Chlorhexidine salts are widely used in pharmaceutical formulations
in Europe and Japan for their antimicrobial properties.
Although mainly used as disinfectants, chlorhexidine salts are also
used as antimicrobial preservatives.
As excipients, chlorhexidine salts are mainly used for the
preservation of eye-drops at a concentration of 0.01% w/v;
generally the acetate or gluconate salt is used for this purpose.
Solutions containing 0.002–0.006% w/v chlorhexidine gluconate
have also been used for the disinfection of hydrophilic contact
lenses.
For skin disinfection, chlorhexidine has been formulated as a
0.5% w/v solution in 70% v/v ethanol and, in conjunction with
detergents, as a 4% w/v surgical scrub. Chlorhexidine salts may also
be used in topical antiseptic creams, mouthwashes, dental gels, and
in urology for catheter sterilization and bladder irrigation.
Chlorhexidine salts have additionally been used as constituents
of medicated dressings, dusting powders, sprays, and creams.
Clinical Use
Chlorhexidine is a biguanide topical antiseptic and disinfectant with broad antimicrobial efficacy. It is increasingly being used as an aseptic but it is also gaining use as a biocidal ingredient in shampoos, conditioners, hair dyes, sunscreens, toothpastes, mouthwashes (Corsodyl), wet wipes (also for babies), eye creams, antiwrinkle creams, moisturizers, contact lens solutions, and instillation gels for urinary catheters.Urticaria following application to intact skin or mucosae, in some cases accompanied by dyspnea, angioedema, syncope, or anaphylaxis has been described via the mucosal route at much lower concentration than elsewhere, generally as low as 0.05%.
Safety Profile
Poison by intraperitoneal andintravenous routes. Mildly toxic by ingestion.Experimental reproductive effects. A human skin irritant.Mutation data reported. When heated to decomposition itemits very toxic fumes of Cl- and NOx.
Incompatibilities
Chlorhexidine salts are cationic in solution and are therefore
incompatible with soaps and other anionic materials. Chlorhexidine
salts are compatible with most cationic and nonionic surfactants,
but in high concentrations of surfactant chlorhexidine activity can
be substantially reduced owing to micellar binding.
Chlorhexidine salts of low aqueous solubility are formed and
may precipitate from chlorhexidine solutions of concentration
greater than 0.05% w/v, when in the presence of inorganic acids,
certain organic acids, and salts (e.g. benzoates, bicarbonates,
borates, carbonates, chlorides, citrates, iodides, nitrates, phosphates,
and sulfates). At chlorhexidine concentrations below
0.01% w/v precipitation is less likely to occur.
In hard water, insoluble salts may form owing to interaction with
calcium and magnesium cations. Solubility may be enhanced by the
inclusion of surfactants such as cetrimide.
Other substances incompatible with chlorhexidine salts include
viscous materials such as acacia, sodium alginate, sodium carboxymethylcellulose,
starch, and tragacanth. Also incompatible
are brilliant green, chloramphenicol, copper sulfate, fluorescein
sodium, formaldehyde, silver nitrate, and zinc sulfate.
Interaction has been reported between chlorhexidine gluconate
and the hydrogel poly(2-hydroxyethyl methacrylate), which is a
component of some hydrophilic contact lenses.
Regulatory Status
Chlorhexidine salts are included in nonparenteral and parenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Chlorhexidine Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Cadila Pharmaceuticals Ltd | +91-7069076657 +91-7069076657 | Ahmedabad, India | 54 | 58 | Inquiry |
| Unilab Company | +91-912228260263 +91-63917864522 | Tamil Nadu, India | 43 | 58 | Inquiry |
| PERFECT CHEMICAL | +91-9820465461 +91-9820465461 | Mumbai, India | 391 | 58 | Inquiry |
| Zuhhad international | +91-8454840801 +91-8454840801 | Mumbai, India | 21 | 58 | Inquiry |
| Meenaakshi Molecules Pvt Ltd | +91-9121311126 +91-9121311126 | Telangana, India | 45 | 58 | Inquiry |
| Dishman Carbogen Amcis Ltd (Dishman Group) | +91-2717420100 +91-8154017093 | Gujarat, India | 90 | 58 | Inquiry |
| Ions Pharma | +912266707500 | Maharashtra, India | 89 | 58 | Inquiry |
| Basic Pharma Life Science Pvt Ltd | +919825056886 | Gujarat, India | 28 | 58 | Inquiry |
| Alex Pharmachem Pvt. Ltd | +91-9825015135 +91-9974615135 | Gujarat, India | 10 | 58 | Inquiry |
| Bajaj Healthcare Ltd | +91-2266177400 +91-2266177400 | Maharashtra, India | 86 | 58 | Inquiry |
Related articles
- The antibacterial properties of chlorhexidine
- Chlorhexidine is an important medical, dental and pharmaceutical antiseptic, disinfectant and preservative.
- Jan 6,2022
55-56-1(Chlorhexidine)Related Search:
1of4
chevron_right




