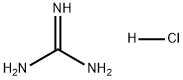
Guanidine hydrochloride
- CAS No.
- 50-01-1
- Chemical Name:
- Guanidine hydrochloride
- Synonyms
- GUANIDINE HCL;GUANIDINIUM CHLORIDE;GACl;GdmCl;GUANIDIUM CHLORIDE;guanidiniumhydrochloride;GUANIDINE HYDROCHLORIDE 99%;Guanidine, hydrochloride (1:1);GUHY;abs9137
- CBNumber:
- CB6677329
- Molecular Formula:
- CH5N3.ClH
- Molecular Weight:
- 95.53
- MOL File:
- 50-01-1.mol
- Modify Date:
- 2024/8/2 16:47:38
| Melting point | 180-185 °C(lit.) |
|---|---|
| Density | 1.18 g/mL at 25 °C(lit.) |
| refractive index |
n |
| storage temp. | room temp |
| solubility | H2O: 6 M, clear, colorless |
| form | Crystals |
| color | White to slightly yellow |
| PH | 4.5-5.5 (100g/l, H2O, 20℃) |
| Odor | Odorless |
| PH Range | 4.5 - 6.0 at 573 g/l at 25 °C |
| Water Solubility | 2280 g/L (20 ºC) |
| Sensitive | Hygroscopic |
| λmax |
λ: 260 nm Amax: 0.040 λ: 280 nm Amax: 0.025 |
| Merck | 14,4562 |
| BRN | 3591990 |
| Stability | Stable. Hygroscopic. Incompatible with strong oxidizing agents. |
| InChIKey | PJJJBBJSCAKJQF-UHFFFAOYSA-N |
| CAS DataBase Reference | 50-01-1(CAS DataBase Reference) |
| EPA Substance Registry System | Guanidine monohydrochloride (50-01-1) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H302+H332-H315-H319 | |||||||||
| Precautionary statements | P261-P264-P301+P312-P302+P352-P304+P340+P312-P305+P351+P338 | |||||||||
| Hazard Codes | Xn,Xi | |||||||||
| Risk Statements | 22-36/38 | |||||||||
| Safety Statements | 26-36-22 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | MF4300000 | |||||||||
| F | 3-10 | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 29252000 | |||||||||
| Toxicity | LD50 orally in Rabbit: 655.3 - 907.1 mg/kg LD50 dermal Rabbit > 2000 mg/kg | |||||||||
| NFPA 704 |
|
Guanidine hydrochloride price More Price(66)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | G4505 | Guanidine hydrochloride ≥99% (titration), organic base and chaeotropic agent | 50-01-1 | 25G | ₹3366.58 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | RDD001 | Guanidine hydrochloride anhydrous, free-flowing, Redi-Dri?, ≥99% | 50-01-1 | 500G | ₹23262.93 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | RDD001 | Guanidine hydrochloride anhydrous, free-flowing, Redi-Dri?, ≥99% | 50-01-1 | 1KG | ₹36718.4 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | G4505 | Guanidine hydrochloride ≥99% (titration), organic base and chaeotropic agent | 50-01-1 | 100G | ₹8789.9 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | G9284 | Guanidine hydrochloride solution | 50-01-1 | 100ML | ₹11528.63 | 2022-06-14 | Buy |
Guanidine hydrochloride Chemical Properties,Uses,Production
Chemical Properties
Guanidine hydrochloride, a strong organic base, appears as white crystal powder or yellowish lumps that are almost insoluble in acetone, benzene, and ether. In physiological pH, it exists primarily as guanidium ions. It is a natural byproduct of protein metabolism found in urine and is commonly used in protein denaturation in laboratory research.
Uses
Guanidine hydrochloride is used for purification of proteins and nucleic acids. It can be used as a medicine, organic synthesis intermediate and is used in dyes. It acts as an intermediate in the preparation of sulfadiazine, which is an important raw material for sulfamethyldiazine, sulfamethazine and folic acid. It is utilized in the synthesis of 2-amino-pyrimidine, 2- amino -6- methyl-pyrimidine and 2-amino -4,6 - dimethyl-pyrimidine. Also used in RNA isolation to dissociate nucleoproteins and inhibit RNase.
Definition
ChEBI: Guanidine Hydrochloride is an aminocarboxamidine, the parent compound of the guanidines. It is a strong organic base existing primarily as guanidium ions at physiological pH. It is found in the urine as a normal product of protein metabolism.
Preparation
The process for preparing guanidine hydrochloride is as follows:
Step 1: Fritting - Combine dicyandiamide and ammonium chloride in a reactor with a 1:1.27 weight ratio and carry out a frit reaction at 170-230°C to obtain crude guanidine hydrochloride.
Step 2: Dissolving - Dissolve the crude guanidine hydrochloride in water at a 1:1 ratio at normal temperature. Then, add ammonium salt alkaloid in an amount determined by the ammonium salt content in the crude guanidine hydrochloride.
Step 3: Filtering - Remove raw materials and reaction by-products with filtration.
Step 4: Dehydrating - Dehydrate the filtered ground mother liquor at a high temperature.
Step 5: Crystallizing - Cool the supersaturated solution, concentrate, and crystallize to obtain high-purity guanidine hydrochloride.
Step 6: Repeat steps 2-5 and add dicyandiamide in the reactor with the guanidine hydrochloride to carry out frit reaction at 170-230°C for 3-4 hours to obtain even higher purity guanidine hydrochloride. This step can be repeated several times if needed.
Step 7: To obtain higher purity guanidine hydrochloride, subject the guanidine hydrochloride obtained in step 5 to steps 2-5 again, with the water ratio in step 2 adjusted to 1.5:1. This step can be repeated numerous times as required.
General Description
Guanidine hydrochloride (GdnHCl) is a well-known protein and enzyme denaturant. It is a small molecule and hydroscopic in nature. It is used to synthesis medicine, pesticide, dye and other organic compound, is the important material to make sulfanilamide medicine and folacin, and can be used as anti-static agent of compound fibre.
Hazard
Toxic by ingestion, evolves hydrogen chloride fumes on heating.
Purification Methods
Crystallise the hydrochloride from hot methanol by chilling to about -10o, with vigorous stirring. The fine crystals are filtered through fritted glass, washed with cold (-10o) methanol, dried at 50o under vacuum for 5hours. (The product is purer than that obtained by crystallisation at room temperature from methanol by adding large amounts of diethyl ether.) [Kolthoff et al. J Am Chem Soc 79 5102 1957, Beilstein 3 H 86, 3 II 71, 3 III 160, 3 IV 150.]
Guanidine hydrochloride Preparation Products And Raw materials
Raw materials
Preparation Products
1of6
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Vihita Chem Private Limited | +91-9512002673 +91-9512002673 | Gujarat, India | 122 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| S D Fine Chem Limited | +91-9323715856 +91-9323715856 | Maharashtra, India | 368 | 58 | Inquiry |
| Trignokem International. | 91-40-27170709 | Andhra Pradesh, India | 233 | 58 | Inquiry |
| Avra Synthesis Pvt Ltd | +914027175796 | New Delhi, India | 1606 | 30 | Inquiry |
| Loba Chemie Pvt Ltd | +91-22-6663 6663 | Mumbai, India | 498 | 58 | Inquiry |
| Sisco Research Laboratories Pvt. Ltd. | +91-22-4268 5800 | Mumbai, India | 4317 | 58 | Inquiry |
| Innovative | 07942565925 | Mumbai, India | 617 | 58 | Inquiry |
| Brisben Chemicals | 08048963750 | Mumbai, India | 509 | 58 | Inquiry |
| Finar Limited | 91-2717-616717 | Gujarat, India | 39 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| Vihita Chem Private Limited | 58 |
| JSK Chemicals | 58 |
| S D Fine Chem Limited | 58 |
| Trignokem International. | 58 |
| Avra Synthesis Pvt Ltd | 30 |
| Loba Chemie Pvt Ltd | 58 |
| Sisco Research Laboratories Pvt. Ltd. | 58 |
| Innovative | 58 |
| Brisben Chemicals | 58 |
| Finar Limited | 58 |
Related articles
- Guanidine Hydrochloride: Therapeutic Effects on Breast Cancer and Health Hazards
- Guanidine hydrochloride shows promise for treating triple-negative breast cancer by targeting KCNG1 gene, but requires careful....
- Jun 26,2024
- The Uses of guanidine hydrochloride
- Guanidine hydrochloride is an organic substance with the chemical formula CH6ClN3, white or yellowish lumps.
- Apr 20,2022
50-01-1(Guanidine hydrochloride)Related Search:
1of4
chevron_right




