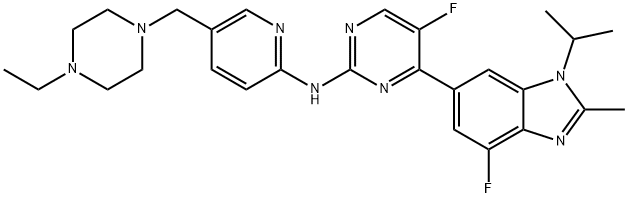
Abemaciclib
- CAS No.
- 1231929-97-7
- Chemical Name:
- Abemaciclib
- Synonyms
- N-[5-[(4-Ethyl-1-piperazinyl)methyl]-2-pyridinyl]-5-fluoro-4-[4-fluoro-2-methyl-1-(1-methylethyl)-1H-benzimidazol-6-yl]-2-pyrimidinamine;Abemaciclib (LY2835219);N-(5-((4-Ethylpiperazin-1-yl)methyl)pyridin-2-yl)-5-fluoro-4-(4-fluoro-1-isopropyl-2-methyl-1H-benzo[d]imidazol-6-yl)pyrimidin-2-amine;CS-814;abecilee;Abemaciclib;Abemaciclib D7;Abemaciclib D10;ABEMACICLIB BASE;Abemaciclib, >=98%
- CBNumber:
- CB72624906
- Molecular Formula:
- C27H32F2N8
- Molecular Weight:
- 506.59
- MOL File:
- 1231929-97-7.mol
- Modify Date:
- 2024/7/2 8:55:16
| Boiling point | 689.3±65.0 °C(Predicted) |
|---|---|
| Density | 1.32±0.1 g/cm3(Predicted) |
| storage temp. | 4°C, protect from light |
| solubility | insoluble in H2O; ≥4.83 mg/mL in DMSO with gentle warming and ultrasonic; ≥6.34 mg/mL in EtOH with gentle warming |
| form | solid |
| pka | 7.69±0.10(Predicted) |
| color | Off-white to yellow |
| InChIKey | UZWDCWONPYILKI-UHFFFAOYSA-N |
| SMILES | C1(NC2=NC=C(CN3CCN(CC)CC3)C=C2)=NC=C(F)C(C2C=C3N(C(C)C)C(C)=NC3=C(F)C=2)=N1 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H373-H360-H410 | |||||||||
| Precautionary statements | P273-P391-P501-P260-P314-P501 | |||||||||
| NFPA 704 |
|
Abemaciclib Chemical Properties,Uses,Production
Characteristics
Class: serine/threonine protein kinase
Treatment: Breast cancer
Oral bioavailability = 46%
Elimination half-life = 18 h
Protein binding = 95–98%
Indications
Abemaciclib is used to treat breast cancer that is estrogen receptor-positive (ER-positive) and HER2-negative. It can be taken by both men and women. Patients may be offered abemaciclib if they have:
Early primary breast cancer that the treatment team thinks has a higher risk of coming back (recurrence). Abemaciclib may be provided as an adjuvant treatment, meaning a treatment given after initial treatment, such as surgery;
Breast cancer that has spread to the tissues and lymph nodes around the chest, neck, and under the breastbone (locally advanced breast cancer);
Breast cancer has spread to other parts of the body, such as the bones, lungs, liver, or brain (secondary breast cancer).
Biochem/physiol Actions
LY2835219 (abemaciclib) was identified via compound and biochemical screening by scientists at Eli Lilly and Company Research Laboratories and selected for its biological activity and highly selective inhibition of the complexes CDK4/ cyclin D1 (IC50 =2 nmol/L) and CDK6/cyclin D1 (IC50 =10 nmol/L), with no activity against other CDK/cyclin complexes or cell-cycle-related kinases within the nanomolar ranges, except for inhibition of CDK9 at IC50 at least five times higher. The compound was shown to act as a competitive inhibitor of the ATP-binding domain of the CDK4 and CDK6 and to be 14 times more potent against CDK4 than against CDK6. In comparison to palbociclib and ribociclib, abemaciclib shows higher selectivity for the complex CDK4/cyclin D1, with IC50 values five times lower than those of the two other compounds[1].
Synthesis
A concise total synthesis of CDK 4/6 inhibitor abemaciclib is described below. The synthesis uses a Suzuki coupling and a Hartwig–Buchwald amination to join three of the four subunits. The final step is a reductive amination utilizing Leuckart–Wallach conditions. Key to the Leuckart–Wallach reaction was the addition of trimethyl orthoformate to remove water formed during the reaction, allowing the reaction to complete[4].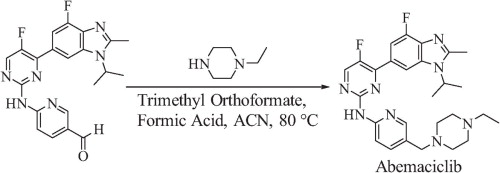
Enzyme inhibitor
This oral cell cycle inhibitor (FWfree-base = 506.61 g/mol; FWmesylate-salt = 602.70 g/mol; CAS 1231930-82-7 (mesylate salt) ), also known as LY2835219 and N-[5-[ (4-ethyl-1-piperazinyl) methyl]-2-pyridinyl]-5- fluoro-4-[4-fluoro-2-methyl-1- (1-methylethyl) -1H-benzimidazol-6-yl]-2- pyrimidinamine, targets the cyclin-dependent kinase CDK4, or cyclin D1 (IC50 = 2 nM) and CDK6, or cyclin D3 (IC50 = 6 nM), inhibiting retinoblastoma (Rb) protein phosphorylation in early G1, thereby arresting the cell cycle in the G1, suppressing DNA synthesis, and inhibiting cancer cell growth. LY2835219 inhibits activation of AKT and ERK, but not mTOR.
References
[1] Silvia Paola Corona, Daniele Generali. “Abemaciclib: a CDK4/6 inhibitor for the treatment of HR+/HER2- advanced breast cancer.” Drug Design, Development and Therapy (2018): 321–330.
[2] “OS Trending Positive for Abemaciclib.” Cancer discovery 13 1 (2023): OF3.
[3] Erin R Scheidemann. “Resistance to abemaciclib is associated with increased metastatic potential and lysosomal protein deregulation in breast cancer cells.” Molecular Carcinogenesis (2024): 209–223.
[4] Michael O. Frederick, Douglas P. Kjell. “A synthesis of abemaciclib utilizing a Leuckart–Wallach reaction.” Tetrahedron Letters 56 7 (2015): Pages 949-951.
Abemaciclib Preparation Products And Raw materials
Raw materials
1of3
chevron_rightPreparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| AVD pharmaceuticals Pvt Ltd | +919860835260 | Pune, India | 102 | 58 | Inquiry |
| Dr. Reddy's Laboratories Ltd | +91-4049002900 +91-4049002900 | Hyderabad, India | 165 | 58 | Inquiry |
| Venkatasai Life Sciences | +91-9908134868 +91-8008303069 | Hyderabad, India | 186 | 58 | Inquiry |
| Apicore Pharmaceuticals Pvt Ltd | +91-2662267177 +91-2662267166 | Gujarat, India | 181 | 58 | Inquiry |
| SynZeal Research Pvt Ltd | +1 226-802-2078 | Gujarat, India | 6522 | 58 | Inquiry |
| Pharma Affiliates | 172-5066494 | Haryana, India | 6761 | 58 | Inquiry |
| MSN LABORATORIES PRIVATE LTD | +91 40 30438600 | New Delhi, India | 230 | 58 | Inquiry |
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| A.J Chemicals | 91-9810153283 | New Delhi, India | 6124 | 58 | Inquiry |
| Pharmaffiliates Analytics and Synthetics P. Ltd | +91-172-5066494 | Haryana, India | 6773 | 58 | Inquiry |
Related articles
- How Abemaciclib works in treating breast cancer
- Preclinical studies have shown that continuous inhibition of CDK4/6 by abemaciclib inhibits phosphorylation of the retinoblast....
- Nov 10,2023
- Abemaciclib: pharmacology, pharmacokinetics and mechanism of action
- Abemaciclib inhibits CDK4/6, blocks cell cycle progression, induces senescence and apoptosis in cancer cells.
- Jul 24,2023
- Uses and Side effects of Abemaciclib
- Abemaciclib (trade names Verzenio and Verzenios) is a drug for the treatment of advanced or metastatic breast cancers. It was ....
- Nov 4,2019





