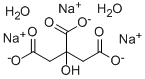
Trisodium citrate dihydrate
- CAS No.
- 6132-04-3
- Chemical Name:
- Trisodium citrate dihydrate
- Synonyms
- CITRIC ACID MONO;SODIUM CITRATE TRIBASIC DIHYDRATE;TRI-SODIUM CITRATE DIHYDRATE;SodiuM Citrate (AS);NATRII CITRAS;SODIUM CITRATE, DIHYDRATE;ACIDUM CITRICUM MONOHYDRICUM;CITRIC ACID TRISODIUM SALT DIHYDRATE;BETA-HYDROXY-TRICARBOXYLIC ACID MONOHYDRATE;abs9147
- CBNumber:
- CB9752065
- Molecular Formula:
- C6H9Na3O9
- Molecular Weight:
- 294.1
- MOL File:
- 6132-04-3.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/7/12 17:58:41
| Melting point | >300 °C(lit.) |
|---|---|
| Density | 1.76 |
| FEMA | 3026 | SODIUM CITRATE |
| Flash point | 173.9 °C |
| storage temp. | Store at +5°C to +30°C. |
| solubility | H2O: 100 mg/mL |
| form | powder |
| color | white |
| Odor | Odorless |
| PH Range | 7.5 - 9 at 29.4 g/l at 25 °C |
| PH | 7.0-9.0 (25℃, 50mg/mL in H2O) |
| Water Solubility | 720 g/L (25 ºC) |
| λmax |
λ: 260 nm Amax: 0.01 λ: 280 nm Amax: 0.01 |
| Merck | 14,8602 |
| BRN | 6104939 |
| Stability | Stable. Incompatible with bases, reducing agents, oxidizing agents. |
| InChIKey | NLJMYIDDQXHKNR-UHFFFAOYSA-K |
| LogP | -1.72 |
| CAS DataBase Reference | 6132-04-3(CAS DataBase Reference) |
| EPA Substance Registry System | 1,2,3-Propanetricarboxylic acid, 2-hydroxy-, trisodium salt, dihydrate (6132-04-3) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS05,GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H318-H319 | |||||||||
| Precautionary statements | P264-P280i-P305+P351+P338-P337+P313-P310a | |||||||||
| Hazard Codes | Xi | |||||||||
| Risk Statements | 37/38-41-36/37/38 | |||||||||
| Safety Statements | 26-36/37/39-24/25-36 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | GE7810000 | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 2918 15 00 | |||||||||
| Toxicity | LD50 orally in Rabbit: > 8000 mg/kg | |||||||||
| NFPA 704 |
|
Trisodium citrate dihydrate price More Price(61)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | W302600 | Sodium citrate dihydrate ≥99%, FG | 6132-04-3 | 1SAMPLE-K | ₹4990.33 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | W302600 | Sodium citrate dihydrate ≥99%, FG | 6132-04-3 | 1KG | ₹7577.5 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | W302600 | Sodium citrate dihydrate ≥99%, FG | 6132-04-3 | 10KG | ₹26542.9 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | W302600 | Sodium citrate dihydrate ≥99%, FG | 6132-04-3 | 25KG | ₹35267.85 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | S4641 | Sodium citrate tribasic dihydrate ACS reagent, ≥99.0% | 6132-04-3 | 25G | ₹1558.8 | 2022-06-14 | Buy |
Trisodium citrate dihydrate Chemical Properties,Uses,Production
Chemical Properties
white powder or colourless crystals
Uses
Trisodium citrate dihydrate, is widely applied in food, beverages and fillers as a buffering, sequestering or an emulsifying agent. It used as an anticoagulant in blood transfusions, osmotic laxative, functional fluids, solvents cleaning, furnishing care products, laundry dishwashing products and cleaning automobile radiators.
Production Methods
Sodium citrate is prepared by adding sodium carbonate to a solution of citric acid until effervescence ceases. The resulting solution is filtered and evaporated to dryness.
Definition
ChEBI: The dihydrate of trisodium citrate.
General Description
Sodium citrate tribasic dihydrate is the tribasic dihydrate sodium salt of citric acid.
Pharmaceutical Applications
Sodium citrate, as either the dihydrate or anhydrous material, is
widely used in pharmaceutical formulations.
It is used in food products, primarily to adjust the pH of
solutions. It is also used as a sequestering agent. The anhydrous
material is used in effervescent tablet formulations. Sodium citrate
is additionally used as a blood anticoagulant either alone or in
combination with other citrates such as disodium hydrogen citrate.
Therapeutically, sodium citrate is used to relieve the painful
irritation caused by cystitis, and also to treat dehydration and
acidosis due to diarrhea.
Biological Activity
Commonly used laboratory reagent
Safety
After ingestion, sodium citrate is absorbed and metabolized to
bicarbonate. Although it is generally regarded as a nontoxic and
nonirritant excipient, excessive consumption may cause gastrointestinal
discomfort or diarrhea. Therapeutically, in adults, up to
15 g daily of sodium citrate dihydrate may be administered orally, in
divided doses, as an aqueous solution to relieve the painful irritation
caused by cystitis.
Citrates and citric acid enhance intestinal aluminum absorption
in renal patients, which may lead to increased, harmful serum
aluminum levels. It has therefore been suggested that patients with
renal failure taking aluminum compounds to control phosphate
absorption should not be prescribed citrate- or citric acid-containing
products.
storage
Sodium citrate dihydrate is a stable material. Aqueous solutions
may be sterilized by autoclaving. On storage, aqueous solutions may cause the separation of small, solid particles from glass
containers.
The bulk material should be stored in an airtight container in a
cool, dry place.
Purification Methods
Crystallise the salt from warm water by cooling to 0o. [Beilstein 3 III 1100, 3 IV 1274.]
Incompatibilities
Aqueous solutions are slightly alkaline and will react with acidic substances. Alkaloidal salts may be precipitated from their aqueous or hydro-alcohol solutions. Calcium and strontium salts will cause precipitation of the corresponding citrates. Other incompatibilities include bases, reducing agents, and oxidizing agents.
Regulatory Status
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (inhalations; injections; ophthalmic products; oral solutions, suspensions, syrups and tablets; nasal, otic, rectal, topical, transdermal, and vaginal preparations). Included in nonparenteral and parenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Trisodium citrate dihydrate Preparation Products And Raw materials
Raw materials
1of2
chevron_rightPreparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Vardhaman P Golechha | 9704422000 | Telangana, India | 294 | 58 | Inquiry |
| Indiana ChemPort | +91-91-2652638667 +91-7574886711 | Gujarat, India | 85 | 58 | Inquiry |
| UNILOSA INTERNATINAL PRIVATE LIMITED | +91-9999069917 +91-9999069917 | New Delhi, India | 152 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| New Alliance Dye Chem Pvt Ltd | +91-9820112338 +91-9820112338 | Maharashtra, India | 104 | 58 | Inquiry |
| Annexe Chem Pvt Ltd | +91-9724624061 +91-9898722162 | Gujarat, India | 57 | 58 | Inquiry |
| Merck Ltd | +91-2262109800 +91-2262109000 | Maharashtra, India | 272 | 58 | Inquiry |
| Kronox Lab Sciences Pvt Ltd | +91-9313231074 +91-9313231074 | Gujarat, India | 240 | 58 | Inquiry |
| India Phosphate | +91-9993319656 +91-9993319656 | Madhya Pradesh, India | 61 | 58 | Inquiry |
| Dhara Industries | +91-9322395199 +91-9322395199 | Mumbai, India | 190 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| Vardhaman P Golechha | 58 |
| Indiana ChemPort | 58 |
| UNILOSA INTERNATINAL PRIVATE LIMITED | 58 |
| JSK Chemicals | 58 |
| New Alliance Dye Chem Pvt Ltd | 58 |
| Annexe Chem Pvt Ltd | 58 |
| Merck Ltd | 58 |
| Kronox Lab Sciences Pvt Ltd | 58 |
| India Phosphate | 58 |
| Dhara Industries | 58 |
6132-04-3(Trisodium citrate dihydrate)Related Search:
1of5
chevron_right




