GERMANIUM(IV) CHLORIDE
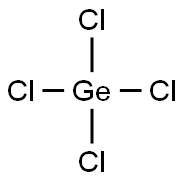
- CAS No.
- 10038-98-9
- Chemical Name:
- GERMANIUM(IV) CHLORIDE
- Synonyms
- GeCl4;ermanium(IV) chL;Germanium(IV) chL;tetrachloro-german;Tetrachlorogermane;GERMANIUM CHLORIDE;tetrachloro-Germane;Germane,tetrachloro-;tetrachloro-germaniu;Germane, tetrachloro-
- CBNumber:
- CB9852574
- Molecular Formula:
- Cl4Ge
- Molecular Weight:
- 214.45
- MOL File:
- 10038-98-9.mol
- Modify Date:
- 2023/11/28 16:31:43
| Melting point | -49.5 °C |
|---|---|
| Boiling point | 83 °C(lit.) |
| Density | 1.844 g/mL at 25 °C |
| vapor pressure | 76 mm Hg ( 20 °C) |
| refractive index | 1.4644 |
| Flash point | 83.1°C |
| storage temp. | Store below +30°C. |
| solubility | Soluble in ether, benzene, chloroform, carbon tetrachloride. Insoluble in hydrochloric acid and sulphuric acid. |
| form | Liquid |
| Specific Gravity | 1.88 |
| color | Colorless |
| PH | <1 (H2O, 20℃) |
| Water Solubility | DECOMPOSES |
| Sensitive | Moisture Sensitive |
| Hydrolytic Sensitivity | 8: reacts rapidly with moisture, water, protic solvents |
| Merck | 14,4410 |
| Dielectric constant | 2.4(25℃) |
| CAS DataBase Reference | 10038-98-9(CAS DataBase Reference) |
| NIST Chemistry Reference | Germanium tetrachloride(10038-98-9) |
| EPA Substance Registry System | Germane, tetrachloro- (10038-98-9) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS05,GHS07,GHS09 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H290-H314-H335-H410 | |||||||||
| Precautionary statements | P234-P261-P273-P280-P303+P361+P353-P305+P351+P338 | |||||||||
| Hazard Codes | C | |||||||||
| Risk Statements | 14-34-67-40-37 | |||||||||
| Safety Statements | 26-27-36-45-8-36/37/39-25 | |||||||||
| RIDADR | UN 3390 6.1/PG 1 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | LY5225000 | |||||||||
| F | 10-19 | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 2827 39 85 | |||||||||
| HazardClass | 8 | |||||||||
| PackingGroup | II | |||||||||
| NFPA 704 |
|
GERMANIUM(IV) CHLORIDE price More Price(4)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 8.04079 | Germanium(IV) chloride for synthesis | 10038-98-9 | 25ML | ₹36370 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 208450 | Germanium(IV) chloride 99.99% trace metals basis | 10038-98-9 | 25G | ₹27701.18 | 2022-06-14 | Buy |
| ottokemi | G 1270 | Germanium(IV) chloride, 99.9999% 99.99% | 10038-98-9 | 25gm | ₹49905 | 2022-05-26 | Buy |
| ottokemi | G 1270 | Germanium(IV) chloride, 99.9999% 99.99% | 10038-98-9 | 100gm | ₹139059 | 2022-05-26 | Buy |
GERMANIUM(IV) CHLORIDE Chemical Properties,Uses,Production
Chemical Properties
Colorless liquid. decomposes in water. Insoluble in concentrated hydrochloric acid; soluble in carbon disulfide, chloroform, benzene, alcohol, and ether.
Physical properties
Colorless liquid; density 1.879 g/cm3 at 20°C and 1.844 g/cm3 at 30°C; refractive index 1.464; boils at 86.5°C; solidifies at -49.5°C; decomposes in water; soluble in alcohol, ether, benzene, chloroform and carbon tetrachloride; insoluble in concentrated hydrochloric and sulfuric acids.
Uses
Germanium(IV) chloride is used in the microwave preparation of Ge2Cl6, a colorless crystalline material which was included in a further study of low-valent germanium compounds.1
Germanium(IV) chloride is used in the preparation of many germanium compounds.
Hazard
Toxic material
Safety Profile
Poison by intravenous route. Mddly toxic by inhalation. A skin, severe eye, and mucous membrane irritant. Will react violently with water or steam to produce toxic and corrosive fumes. When heated to decomposition it emits toxic fumes of Cl-. See also GERMANIUM COMPOUNDS.
Purification Methods
Traces of Cl2 and HCl can be removed from the liquid by blowing dry air through it for a few hours at room temperature or by shaking it with Hg or Hg2Cl2 and then fractionating it in a vacuum. It decomposes on heating at 950o. It has a sharp penetrating odour and fumes in moist air to give a chalky coat of GeO2. It is slowly hydrolysed by H2O to give GeO2, but distils from conc HCl. [Foster et al. Inorg Synth II 109 1946, Dennis & Hance J Am Chem Soc 44 304 1922, Schenk in Handbook of Preparative Inorganic Chemistry (Ed. Brauer) Academic Press Vol I p 715 1963.] LACHRYMATORY. Glass powder (100-300 mesh). Washed with 10% HNO3, water and dry in air.
GERMANIUM(IV) CHLORIDE Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| Otto Chemie Pvt. Ltd. | +91 9820041841 | Mumbai, India | 5873 | 58 | Inquiry |
| Chemik Chemicals | 08066085906Ext 278 | Gujarat, India | 10 | 58 | Inquiry |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | China | 21663 | 55 | Inquiry |
| Chongqing Chemdad Co., Ltd | +86-023-6139-8061 +86-86-13650506873 | China | 39916 | 58 | Inquiry |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418671 +8618949823763 | China | 34571 | 58 | Inquiry |
| Dayang Chem (Hangzhou) Co.,Ltd. | 571-88938639 +8617705817739 | China | 52861 | 58 | Inquiry |
| CD Chemical Group Limited | +8615986615575 | China | 20356 | 58 | Inquiry |
| Shaanxi Didu New Materials Co. Ltd | +86-89586680 +86-13289823923 | China | 8672 | 58 | Inquiry |
| PT CHEM GROUP LIMITED | China | 35426 | 58 | Inquiry |
Related articles
- Preparations and Reaction of Germanium tetrachloride
- Germanium(IV) chloride often is obtained as a byproduct of germanium metal production. The process involves heating germanium ....
- Oct 21,2019





