Ferrous chloride
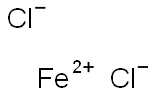
- CAS No.
- 7758-94-3
- Chemical Name:
- Ferrous chloride
- Synonyms
- FECL2;IRON CHLORIDE;IRON(II) CHLORIDE;Ferrofloc;FERROUS CHLORIDE ANHYDROUS;Iron chloride (FeCl2);Iron(II) chloride anhydrous;ferro66;NA 1759;Ferro 66
- CBNumber:
- CB5411955
- Molecular Formula:
- Cl2Fe
- Molecular Weight:
- 126.75
- MOL File:
- 7758-94-3.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/3/14 15:18:26
| Melting point | 677 °C (lit.) |
|---|---|
| Boiling point | 1023°C |
| Density | 3.16 g/mL at 25 °C (lit.) |
| vapor pressure | 0Pa at 20℃ |
| Flash point | 1023°C |
| solubility | H2O: soluble |
| form | beads |
| color | Off-white |
| Specific Gravity | 3.162 |
| Water Solubility | Soluble in water, alcohol and acetone. Slightly soluble in benzene. Insoluble in ether. |
| Sensitive | Hygroscopic |
| Merck | 14,4043 |
| Exposure limits |
ACGIH: TWA 1 mg/m3 NIOSH: TWA 1 mg/m3 |
| InChIKey | NMCUIPGRVMDVDB-UHFFFAOYSA-L |
| CAS DataBase Reference | 7758-94-3(CAS DataBase Reference) |
| NIST Chemistry Reference | Iron dichloride(7758-94-3) |
| EPA Substance Registry System | Ferrous chloride (7758-94-3) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS05,GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H302-H318 | |||||||||
| Precautionary statements | P264-P270-P280-P301+P312-P305+P351+P338-P501 | |||||||||
| Hazard Codes | C | |||||||||
| Risk Statements | 22-34 | |||||||||
| Safety Statements | 26-36/37/39-45 | |||||||||
| RIDADR | UN 3260 8/PG 3 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | NO5400000 | |||||||||
| F | 3-10-23 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 8 | |||||||||
| PackingGroup | III | |||||||||
| HS Code | 2827392000 | |||||||||
| NFPA 704 |
|
Ferrous chloride price More Price(18)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 450944 | Iron(II) chloride AnhydroBeads?, ?10?mesh, 99.9% trace metals basis | 7758-94-3 | 10G | ₹10987.38 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 450944 | Iron(II) chloride AnhydroBeads?, ?10?mesh, 99.9% trace metals basis | 7758-94-3 | 50G | ₹35181.25 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 450936 | Iron(II) chloride AnhydroBeads?, ?10?mesh, 99.99% trace metals basis | 7758-94-3 | 5G | ₹10716.75 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 450936 | Iron(II) chloride AnhydroBeads?, ?10?mesh, 99.99% trace metals basis | 7758-94-3 | 25G | ₹25211.43 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 429368 | Iron(II) chloride AnhydroBeads?, ?10?mesh, 99.998% trace metals basis | 7758-94-3 | 1G | ₹6137.78 | 2022-06-14 | Buy |
Ferrous chloride Chemical Properties,Uses,Production
Chemical Properties
Ferrous chloride is a pale greenish salt-like crystal or power.
Physical properties
White hexagonal crystal; hygroscopic; density 3.16g/cm3; melts at 677°C;vaporizes at 1,023°C; vapor pressure 20 torr at 737°C and 200 torr at 897°C;highly soluble in water, ethanol and acetone; slightly soluble in benzene. Thedihydrate and tetrahydrate are greenish monoclinic crystals; densities 2.39and 1.39 g/cm3, respectively; decomposing at 120 and 105°C, respectively;both the hydrates soluble in water.
Uses
Ferrous chloride (FeCl2) is used in pharmaceutical preparations, for sewage treatment, and as a mordant (which fixes dyes so that they will not run) in textiles.
Preparation
Iron(II) chloride is prepared by passing chlorine or hydrogen chloride gasover iron at red heat or 700°C:
Fe + 2HCl → FeCl2 + H2
Fe + Cl2 → FeCl2
It also may be produced by the reduction of iron(III) chloride with hydrogenor other reducing agents at elevated temperatures:
2FeCl3 + H2 → 2FeCl2 + 2HCl
The tetrahydrate is obtained by dissolving the metal in hydrochloric acidfollowed by crystallization at room temperature.
Fe + 2HCl + 4H2O → FeCl2•4H2O + H2
The tetrahydrate gradually loses water when heated above 105°C formingdihydrate, monohydrate and the anhydrous salt. At 220°C it loses all its waterof crystallization.
General Description
Ferrous chloride is a greenish white crystalline solid. Ferrous chloride is soluble in water. Ferrous chloride is noncombustible. Ferrous chloride is used in sewage treatment, in dyeing of fabrics, and for many other uses.
Air & Water Reactions
Water soluble.
Reactivity Profile
Alkali metal hydroxides, acids, anhydrous chlorides of iron, tin, and aluminum, pure oxides of iron and aluminum, and metallic potassium are some of the catalysts that may cause ethylene oxide to rearrange and polymerize, liberating heat, [J. Soc. Chem. Ind. 68:179(1949)]. Explosions occur , although infrequently, from the combination of ethylene oxide and alcohols or mercaptans, [Chem. Eng. News 20:1318(1942)].
Health Hazard
Inhalation of dust irritates nose and throat. Ingestion causes irritation of mouth and stomach. Dust irritates eyes and may cause skin irritation on prolonged contact.
Fire Hazard
Special Hazards of Combustion Products: Irritating hydrogen chloride fumes may form in fire.
Safety Profile
Poison by ingestion and intraperitoneal routes. Mutation data reported. Corrosive. Probably an irritant to the eyes, skin, and mucous membranes. Can react violently with ethylene oxide, K, Na. di%en heated to decomposition it emits toxic fumes of Cl-. See also CHLORIDES and IRON.
Potential Exposure
It is used in textile dyeing, metallurgy, the pharmaceutical industry and sewage treatment.
Shipping
UN1759 Ferrous chloride, solid, Hazard class: 8; Labels: 8-Corrosive material. UN1760 Ferrous chloride, solution, Hazard class: 8; Labels: 8-Corrosive material.
Purification Methods
It forms white hygroscopic rhombohedral crystals with a green tint which oxidise in air to FeCl3 and Fe2O3. It is soluble in H2O, EtOH Me2CO but insoluble in Et2O. The tetrahydrate is pale green to pale blue in colour and loses 2H2O at 105-115o. The dihydrate loses H2O at 120o. [Anhydrous FeBr2 can be obtained by carefully dehydrating the tetrahydrate in a stream of HBr and N2, and it can be sublimed under N2.] The ferrous iron in aqueous solutions of these salts readily oxidises to ferric iron. (See above.) [Kovacuumic & Brace Inorg Synth VI 172 1960, Lux in Handbook of Preparative Inorganic Chemistry (Ed Brauer) Academic Press Vol II p 1491 1965.]
Incompatibilities
Solution attacks metals. Contact with ethylene oxide may initiate polymerization. Contact with potassium or sodium forms an impact-sensitive material.
Ferrous chloride Preparation Products And Raw materials
Raw materials
Preparation Products
1of3
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Yogi Dye Chem Industries | +91-9821098577 +91-9821098577 | Mumbai, India | 124 | 58 | Inquiry |
| Nithyasri Chemicals (APURVA CHEMICALS) | +91-2512692130 +91-9323652199 | Maharashtra, India | 75 | 58 | Inquiry |
| A.J Chemicals | 91-9810153283 | New Delhi, India | 6124 | 58 | Inquiry |
| Otto Chemie Pvt. Ltd. | +91 9820041841 | Mumbai, India | 5873 | 58 | Inquiry |
| Alfa Aesar | 1 800 209 7001 | Maharashtra, India | 6913 | 58 | Inquiry |
| Advance Bio Science | 07942558032 | Gujarat, India | 5 | 58 | Inquiry |
| Nithyasri Chemicals | 08046044427 | Maharashtra, India | 43 | 58 | Inquiry |
| Babu Ram & Sons | 08048372485Ext 470 | Delhi, India | 2 | 58 | Inquiry |
| Hebei Mojin Biotechnology Co., Ltd | +86 13288715578 +8613288715578 | China | 12460 | 58 | Inquiry |
| Hebei Guanlang Biotechnology Co,.LTD | +86-19930503253; +8619930503252 | China | 5911 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| Yogi Dye Chem Industries | 58 |
| Nithyasri Chemicals (APURVA CHEMICALS) | 58 |
| A.J Chemicals | 58 |
| Otto Chemie Pvt. Ltd. | 58 |
| Alfa Aesar | 58 |
| Advance Bio Science | 58 |
| Nithyasri Chemicals | 58 |
| Babu Ram & Sons | 58 |
| Hebei Mojin Biotechnology Co., Ltd | 58 |
| Hebei Guanlang Biotechnology Co,.LTD | 58 |
7758-94-3(Ferrous chloride)Related Search:
1of4
chevron_right




