Nickel chloride
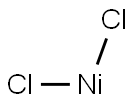
- CAS No.
- 7718-54-9
- Chemical Name:
- Nickel chloride
- Synonyms
- NiCl2;NiCl;NICKEL DICHLORIDE;NICKEL(II) CHLORIDE;Hexahydrate;LiCl2.6H2O;dichloronickel;NICKEL CHLORIDE;Nichel chloride;NICKEL(+2)CHLORIDE
- CBNumber:
- CB0116468
- Molecular Formula:
- Cl2Ni
- Molecular Weight:
- 129.6
- MOL File:
- 7718-54-9.mol
- MSDS File:
- SDS
- Modify Date:
- 2023/9/22 14:52:15
| Melting point | 1001 °C |
|---|---|
| Boiling point | 987°C |
| Density | 3.55 g/mL at 25 °C(lit.) |
| vapor pressure | 1.33 hPa (671 °C) |
| storage temp. | Store below +30°C. |
| solubility | H2O: soluble |
| form | powder |
| Specific Gravity | 3.55 |
| color | Yellow to orange |
| PH | 4 (500g/l, H2O, 20℃) |
| Water Solubility | slightly soluble |
| Sensitive | Hygroscopic |
| Merck | 14,6505 |
| Exposure limits |
ACGIH: TWA 0.1 mg/m3 NIOSH: IDLH 10 mg/m3; TWA 0.015 mg/m3 |
| Stability | Stable. Incompatible with peroxides. |
| InChIKey | QMMRZOWCJAIUJA-UHFFFAOYSA-L |
| CAS DataBase Reference | 7718-54-9(CAS DataBase Reference) |
| NIST Chemistry Reference | Nickel dichloride(7718-54-9) |
| EPA Substance Registry System | Nickel(II) chloride (7718-54-9) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS06,GHS08,GHS09 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H301+H331-H315-H317-H334-H341-H350i-H360D-H372-H410 | |||||||||
| Precautionary statements | P201-P273-P280-P301+P310-P302+P352-P304+P340+P311 | |||||||||
| Hazard Codes | T,N,Xi | |||||||||
| Risk Statements | 45-25-36/38-43-50-50/53-22-52/53-68-51/53-48/23-42/43-20/22-61-49-38-23/25 | |||||||||
| Safety Statements | 53-26-36/37-45-61-37-29-24-23-60 | |||||||||
| RIDADR | UN 3288 6.1/PG 3 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | QR6475000 | |||||||||
| F | 3 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 6.1(b) | |||||||||
| PackingGroup | III | |||||||||
| HS Code | 28273500 | |||||||||
| Toxicity | LD50 in mice, rats (mg/kg): 48, 11 i.p. (IARC) | |||||||||
| NFPA 704 |
|
Nickel chloride price More Price(16)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 8.06722 | Nickel(II) chloride anhydrous for synthesis | 7718-54-9 | 100G | ₹11979.99 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 451193 | Nickel(II) chloride anhydrous, powder, 99.99% trace metals basis | 7718-54-9 | 5G | ₹12849.28 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 451193 | Nickel(II) chloride anhydrous, powder, 99.99% trace metals basis | 7718-54-9 | 25G | ₹39175.68 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 339350 | Nickel(II) chloride 98% | 7718-54-9 | 50G | ₹2673.78 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 339350 | Nickel(II) chloride 98% | 7718-54-9 | 250G | ₹10478.6 | 2022-06-14 | Buy |
Nickel chloride Chemical Properties,Uses,Production
Description
Nickel chloride, NiCl2, is a water-soluble salt used in a variety of industries. It is typically produced and used either as an anhydrous salt or as a hydrate: nickel chloride hexahydrate as well as nickel chloride dihydrate. Nickel chloride is very rarely found in nature, and is produced via extraction from nickelcontaining ores via treatment with hydrochloric acid.
Chemical Properties
Nickel chloride appears as green or brown scales, or sparkling golden-yellow powder.
Physical properties
The anhydrous salt forms yellow crystal scales; deliquesces; density 3.55 g/cm3; melts at 1,001°C; sublimes at 973°C; highly soluble in water, 64 g/100mL at 20°C; soluble in alcohol.
The hexahydrate forms green monoclinic crystals; deliquesces; extremely soluble in water, 254 g/100mL at 20°C, and about 600 g/100 ml at 100°C; also very soluble in alcohol.
Uses
Nickel chloride solutions are used for electroplating nickel onto other metal items.It also is used to prepare various nickel salts and nickel catalysts; and in industrial gas masks to protect from ammonia.
Preparation
Nickel(II) chloride can be obtained by reaction of the elements either in a flow system at high temperatures or by reaction in ethanol at 20°. It is readily prepared in the laboratory by dehydration of the hexahydrate with thionyl chloride.
Production Methods
Nickel chloride (hexahydrate) is obtained by reacting metal nickel powder or nickel oxide with hot, dilute hydrochloric acid.
Definition
ChEBI: A compound of nickel and chloride in which the ratio of nickel (in the +2 oxidation state) to chloride is 1:2.
Nickel chloride is a yellow deliquescent solid with a boiling point of 973°C(1690°F). Nickel chloride is soluble in water and alcohol. Nickel chloride(hydrated),NiCI2·H20, is a gray deliquescent solid that is also soluble in water and alcohol.It is used in nickel plating.
General Description
Nickel(II) chloride (NiCl2) is a nickel based halide that is prepared by burning nickel in chlorine. It is a water soluble compound that crystallizes to form a hexahydrate. It is majorly utilized in organic synthesis as a catalyst and a precursor.
Hazard
Confirmed carcinogen.
Safety Profile
Confirmed human carcinogen. Poison by ingestion, intravenous, intramuscular, and intraperitoneal routes. An experimental teratogen. Experimental reproductive effects. Mutation data reported. When heated to decomposition it emits very toxic fumes of Cl-. See also NICICEL COMPOUNDS.
Potential Exposure
Nickel chloride is used in electroplating and ink manufacturing.
Environmental Fate
Nickel chloride is water soluble (642 g l-1 for anhydrous;
2540 g l-1 for hexahydrate) and would be expected to release
divalent nickel into the water. Since nickel chloride quickly
dissolves upon exposure to moist environments, and partially
due to the ubiquity of nickel in soil, water, and air, tracking the
course of the salt through the environment is difficult. This is
particularly due to nickel’s ability to complex with anionic
species other than chloride to form nickel oxide, sulfate, nitrate,
carbonate, or acetate, among others.
Industrial uses of nickel chloride result in nickel being
distributed mainly at soil surfaces and through surrounding
waterways and water tables. Once distributed to the soil, nickel
chloride produces nickel(II) ions to potentially form inorganic
crystalline minerals or precipitates, can complex or adsorb onto
organic and inorganic surfaces, can participate in cation
exchange, and can exist as free-ion or chelated metal complexes
in soil solution.
Shipping
UN3288 Toxic solids, inorganic, n.o.s., Hazard Class: 6.1; Labels: 6.1-Poisonous materials, Technical Name Required.
Purification Methods
It crystallises from dilute HCl to form the green hexahydrate. At 70o this dehydrates to the tetrahydrate, and at higher temperatures it forms the anhydrous salt. It sublimes in yellow hexagonal scales in a stream of HCl. Store it in a desiccator as it is deliquescent. [Hart & Partington J Chem Soc 104 1943.]
Incompatibilities
Strong acids, potassium, sulfur. Forms an impact-sensitive mixture with potassium.
Waste Disposal
Recycle or disposal in a chemical waste landfill is recommended.
Nickel chloride Preparation Products And Raw materials
Raw materials
1of2
chevron_rightPreparation Products
1of2
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| GLR Innovations | +91 9891111994 | New Delhi, India | 4542 | 58 | Inquiry |
| JAGGO OVERSEAS | +91-9999536088 +91-9999536089 | New Delhi, India | 6 | 58 | Inquiry |
| Kronox Lab Sciences Pvt Ltd | +91-9313231074 +91-9313231074 | Gujarat, India | 240 | 58 | Inquiry |
| Sajan Overseas Pvt Ltd | +91-9825074205 +91-9825074205 | Gujarat, India | 38 | 58 | Inquiry |
| Delcra Chemicals | +91-2226852116 +91-9820047783 | Maharashtra, India | 11 | 58 | Inquiry |
| Shriji Chemicals | +91-9822328437 +91-9822328437 | Maharashtra, India | 14 | 58 | Inquiry |
| Shinde Chemicals Pvt Ltd | +91-9930889810 +91-9930889810 | Maharashtra, India | 15 | 58 | Inquiry |
| Kezi Industries (Sam Industries) | +91-9426862414 +91-9825269675 | Gujarat, India | 34 | 58 | Inquiry |
| Monopoly Innovations Pvt. Ltd. | +undefined-9321425897 +undefined-9819745149 | Maharashtra, India | 45 | 58 | Inquiry |
| Ultimate Chem (India) Pvt., Ltd. | 91-22-29201582 | Maharashtra, India | 117 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| GLR Innovations | 58 |
| JAGGO OVERSEAS | 58 |
| Kronox Lab Sciences Pvt Ltd | 58 |
| Sajan Overseas Pvt Ltd | 58 |
| Delcra Chemicals | 58 |
| Shriji Chemicals | 58 |
| Shinde Chemicals Pvt Ltd | 58 |
| Kezi Industries (Sam Industries) | 58 |
| Monopoly Innovations Pvt. Ltd. | 58 |
| Ultimate Chem (India) Pvt., Ltd. | 58 |





