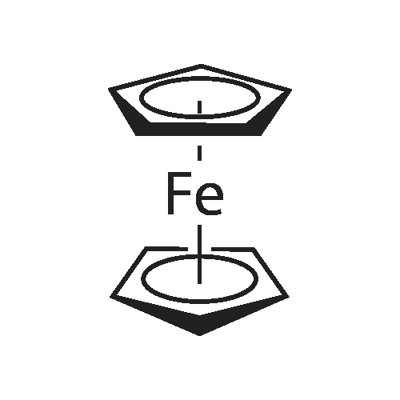
Ferrocene
- CAS No.
- 102-54-5
- Chemical Name:
- Ferrocene
- Synonyms
- ermaotie;FERROCEN;catane;Oxolan-3-ol;DICYCLOPENTADIENYLIRON;BIS(CYCLOPENTADIEN)IRON;IRON DICYCLOPENTADIENYL;BIS(CYCLOPENTADIENYL)IRON;FERROCENE;Ferrotsen
- CBNumber:
- CB1414721
- Molecular Formula:
- C10H10Fe
- Molecular Weight:
- 186.03
- MOL File:
- 102-54-5.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/9/5 22:20:32
| Melting point | 172-174 °C (lit.) |
|---|---|
| Boiling point | 249 °C (lit.) |
| Density | 1.490 |
| vapor pressure | 0.03 mm Hg ( 40 °C) |
| Flash point | 100°C |
| storage temp. | Store below +30°C. |
| solubility | insoluble in H2O; soluble in ethanol, ethyl ether,benzene, dilute HNO 3 |
| form | crystal |
| color | orange |
| Water Solubility | practically insoluble |
| Sensitive | Air & Moisture Sensitive |
| Sublimation | 100 ºC |
| Merck | 14,4037 |
| Exposure limits |
ACGIH: TWA 10 mg/m3; TWA 1 mg/m3 OSHA: TWA 15 mg/m3; TWA 5 mg/m3 NIOSH: TWA 10 mg/m3; TWA 5 mg/m3; TWA 1 mg/m3 |
| Stability | Stable at room temperature. Incompatible with strong oxidizing agents. Highly flammable. |
| LogP | 3.711 at 22℃ |
| CAS DataBase Reference | 102-54-5(CAS DataBase Reference) |
| NIST Chemistry Reference | Ferrocene(102-54-5) |
| EPA Substance Registry System | Ferrocene (102-54-5) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |     GHS02,GHS07,GHS08,GHS09 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H228-H302+H332-H360FD-H373-H410 | |||||||||
| Precautionary statements | P202-P210-P273-P301+P312-P304+P340+P312-P308+P313 | |||||||||
| Hazard Codes | F,Xn,N | |||||||||
| Risk Statements | 11-22-51/53-2017/11/22 | |||||||||
| Safety Statements | 61-22-24/25 | |||||||||
| RIDADR | UN 1325 4.1/PG 2 | |||||||||
| OEB | B | |||||||||
| OEL | TWA: 10 mg/m3 (total) | |||||||||
| WGK Germany | 2 | |||||||||
| RTECS | LK0700000 | |||||||||
| Autoignition Temperature | >150 °C | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 4.1 | |||||||||
| PackingGroup | II | |||||||||
| HS Code | 29310095 | |||||||||
| Toxicity | LD50 orally in Rabbit: 1320 mg/kg LD50 dermal Rat > 3000 mg/kg | |||||||||
| NFPA 704 |
|
Ferrocene price More Price(18)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | F408 | Ferrocene 98% | 102-54-5 | 5G | ₹1948.5 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | F408 | Ferrocene 98% | 102-54-5 | 100G | ₹2468.1 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | F408 | Ferrocene 98% | 102-54-5 | 500G | ₹8205.35 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 8.03978 | Ferrocene for synthesis | 102-54-5 | 50G | ₹3300 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 8.03978 | Ferrocene for synthesis | 102-54-5 | 5KG | ₹52450.01 | 2022-06-14 | Buy |
Ferrocene Chemical Properties,Uses,Production
Chemical Properties
Ferrocene, a metallocene, is a bright orange salt-like crystals from alcohol. Camphor odor.
Physical properties
Orange crystals; camphor-like odor; melts at 172.5°C; vaporizes at 249°C; sublimes above 100°C; thermally stable above 500°C; insoluble in water; soluble in alcohol, ether and benzene; also soluble in dilute nitric acid and concentrated sulfuric acid forming a deep red solution that fluoresces.
Uses
Ferrocene is used as a catalyst for vulcanization, acceleration, and polymerization, as a chemical intermediate for polymeric compounds such as high temperature polymers, as an antiknock additive for gasoline, as a coating for missiles and satellites, and as a high-temperature lubricant.
Production Methods
Ferrocene is produced from the reaction of cyclopentadiene with reduced iron in the presence of metal oxides. There is also a two-stage production process in which produced iron (II)oxide (stage 1) is reacted with cyclopentadiene.
Definition
ferrocene: An orange-red crystallinesolid, Fe(C5H5)2; m.p. 173°C. Itcan be made by adding the ioniccompound Na+C5H5- (cyclopentadienylsodium, made from sodium andcyclopentadiene) to iron(III) chloride.In ferrocene, the two rings are parallel,with the iron ion sandwiched betweenthem (hence the namesandwich compound: see formula).The bonding is between pi orbitalson the rings and d-orbitals on theFe2+ ion. The compound can undergoelectrophilic substitution on theC5H5rings (they have some aromatic character).It can also be oxidized to theblue ion (C5H5)2Fe+. Ferrocene is the first of a class of similar complexescalled sandwich compounds. Its systematicname is di-π-cyclopentadienyliron(II).
Preparation
Dicyclopentadienyliron may be obtained in a single-step synthetic route by heating cyclopentadiene with iron or iron pentacarbonyl at 300°C:
2C5H5 + Fe → (C5H5)2Fe
Also, it can be prepared by the reaction of iron(II) chloride with cyclopentadiene in the presence of an alkyl amine or a similar base.
Another convenient method of preparing this π-complex of iron is a twostep process in which the first step involves preparation of cyclopentadienyl Grignard reagent, such as 2,4-cyclopentadienylmagnesium bromide C5H5MgBr which may then be combined with ferric chloride to yield dicyclopentadienyl iron:
3C5H5MgBr + FeCl3 → (C5H5)2Fe + 3MgBrCl
Another general method of preparation involves the reaction of cyclopentadiene with sodium metal or sodium hydride in tetrahydrofuran (THF). Addition of iron(II) chloride to this solution forms the complex dicyclopentadienyliron:
2C5H6 + 2Na → 2C5H5ˉ + 2Na+ + H2
In 3:2 molar ratio of cyclopentadiene to sodium cyclopentene is obtained along with cyclopentadienidide (C5H5ˉ ) anion:
3C5H6 + 2Na → 2C5H5¯ + 2Na+ + C5H8
FeCl2 + 2C5H6Na → (C5H5)2Fe + 2NaCl
General Description
Orange crystalline solid or orange-yellow powder. Sublimes above 212°F. Camphor odor.
Air & Water Reactions
Sensitive to prolonged exposure to air and may be sensitive to light. Insoluble in water.
Reactivity Profile
Ferrocene reacts violently with tetranitromethane. . Contact of tetranitromethane with Ferrocene under various conditions leads to violent explosion, [Trans. Met. Chem., 1979, 4, 207-208].
Hazard
Moderate fire risk. Evolves toxic products on decomposition and heating.
Health Hazard
Dicyclopentadienyl iron causes
changes in blood parameters and hepatic
cirrhosis.
The toxicological properties of dicyclopentadienyl
iron have not been extensively
investigated. However, it has been used as a
preventive and therapeutic iron deficiency
drug, and its utilization is listed as tolerable.
Fire Hazard
Flash point data for Ferrocene are not available. Ferrocene is probably combustible.
Safety Profile
Poison by intraperitoneal and intravenous routes. Moderately toxic by ingestion. Questionable carcinogen with experimental tumorigenic data. Mutation data reported. Flammable; reacts violently with NH4ClO4. When heated to decomposition it emits acrid smoke and irritating fumes.
Potential Exposure
Used as additive in fuel oil; antiknock agent in gasoline fuel; used in making rubber, silicone resins, high-temperature polymers and lubricants; interme diate for high-temperature polymers; as a smoke suppres sant and catalyst
Carcinogenicity
Ferrocene was administered by intramuscular injection at a dose of 5175 mg/kg/2 years. By the criterion established by the Registry of Toxic Effects of Chemical Substances (RTECS), ferrocene was an equivocal tumorigenic agent and tumors were most evident at the site of multiple injections.
Shipping
UN1325 Flammable solids, organic, n.o.s., Hazard Class: 4.1; Labels: 4.1-Flammable solid.
Purification Methods
Purify it by crystallisation from pentane or cyclohexane (also *C6H6 or MeOH can be used). It is moderately soluble in Et2O and sublimes readily above 100o. Crystallisation from EtOH gave material m 172.5-173o. [Wilkinson Org Synth Coll Vol IV 473 1963, Miller J Chem Soc 632 1952.] It has also been crystallised from methanol and sublimed in vacuo. [Saltiel et al. J Am Chem Soc 109 1209 1987, Beilstein 16 IV 1783.]
Incompatibilities
Violent reaction with ammonium per chlorate, tetranitromethane, mercury(II) nitrate. Incompa tible with oxidizers (chlorates, nitrates, peroxides, perman ganates, perchlorates, chlorine, bromine, fluorine, etc.);
contact may cause fires or explosions. Keep away from
alkaline materials, strong bases, strong acids, oxoacids,
epoxides.
Peroxomonosulfuric acid. Decomposes @≧465 ℃.
Ferrocene Preparation Products And Raw materials
Raw materials
1of2
chevron_rightPreparation Products
1of4
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| GLR Innovations | +91 9891111994 | New Delhi, India | 4542 | 58 | Inquiry |
| SNA HEALTHCARE PVT LTD | +91-8652842416 +91-8652842416 | Mumbai, India | 88 | 58 | Inquiry |
| Dr. Silviu Pharmachem Pvt., Ltd. | +91-8390608382 +91-8390608382 | Mumbai, India | 248 | 58 | Inquiry |
| Oxford laboratories | +91 250 239 0032 | Maharashtra, India | 652 | 58 | Inquiry |
| Spectrochem Private Limited | 08048372608Ext 246 | Maharashtra, India | 1056 | 58 | Inquiry |
| Anand Agencies | 91-20-24454597 | Maharashtra, India | 2337 | 58 | Inquiry |
| Dr. Silviu Pharmachem Pvt., Ltd. (DRSPL) | 91-9665522151 | Maharashtra, India | 143 | 58 | Inquiry |
| Laurice Labs | 08048372498Ext 403 | Mumbai, India | 58 | 58 | Inquiry |
| Sisco Resech Laboratories Private Limited | +91-22-4268 5800 | Mumbai, India | 655 | 58 | Inquiry |
| Otto Chemie Pvt. Ltd. | +91 9820041841 | Mumbai, India | 5873 | 58 | Inquiry |
Related articles
- Ferrocene: An Iconic Organometallic Compound
- Ferrocene, one of the most iconic compounds in the field of organometallic chemistry, has fascinated chemists since its discov....
- Jul 12,2024
- Application of ferrocene in chemical reactions
- Ferrocene is an organic transition metal compound with aromatic properties. Its chemical formula is Fe(C5H5)2.
- Apr 8,2022
102-54-5(Ferrocene)Related Search:
1of4
chevron_right




