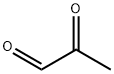
메틸 글리옥살 C화학적 특성, 용도, 생산
화학적 성질
clear yellow to yellow-brown solution
출처
Reported found in the dry distillate of Manilla copal. Also reported found in apple juice, orange juice, celery root, rutabaga, tomato, wheaten bread, white bread, roasted and raw turkey, cognac, roasted barley, beer, cocoa, coffee and roasted pecans.
용도
Methylglyoxal solution has been used:
- to assess glyoxalase 1 (GLO1) enzymatic activity
- as an advanced glycation end (AGE) forming agent for the preparation of albumin in vitro
- to regulate anxiety like behavior in mice
- to induce peritoneal fibrosis in rats
- to study the chromatographic retention characteristics of organic chemicals and metal DNA adducts
- for intraplantar injection in mice to investigate peripheral and central components of methylglyoxal (MG)-transient receptor potential ankyrin 1 (TRPA1)-adenylyl cyclase 1 isoform (AC1) pathway
제조 방법
By distilling a dilute solution of dihydroxyacetone from calcium carbonate; by oxidation of acetone with selenium dioxide; by heating dihydroxy acetone with phosphorus pentoxide; by warming isonitroso acetone with diluted H2SO4.
정의
ChEBI: A 2-oxo aldehyde derived from propanal.
일반 설명
Clear yellow slightly viscous liquid with a pungent odor. Yellowish-green vapors. Faintly acidic to litmus.
공기와 물의 반응
Water soluble.
반응 프로필
Methylglyoxal polymerizes readily. Methylglyoxal is hygroscopic. Methylglyoxal is incompatible with strong oxidizing agents and bases. Methylglyoxal is an aldehyde. Aldehydes are frequently involved in self-condensation or polymerization reactions. These reactions are exothermic; they are often catalyzed by acid. Aldehydes are readily oxidized to give carboxylic acids. Flammable and/or toxic gases are generated by the combination of aldehydes with azo, diazo compounds, dithiocarbamates, nitrides, and strong reducing agents. Aldehydes can react with air to give first peroxo acids, and ultimately carboxylic acids. These autoxidation reactions are activated by light, catalyzed by salts of transition metals, and are autocatalytic (catalyzed by the products of the reaction). The addition of stabilizers (antioxidants) to shipments of aldehydes retards autoxidation.
화재위험
Literature sources indicate that Methylglyoxal is nonflammable.
환경귀착
Methylglyoxal production and use as a chemical intermediate
and flavoring agent may result in its release to the environment
through various waste streams. If released into water, MG is not
expected to adsorb to suspended solids and sediment based on
the estimated Koc. Volatilization from water surfaces is not
expected to be an important fate process based upon the estimated
Henry’s Law constant. If released to soil, MG is expected
to have very high mobility based upon an estimated Koc of 1
determined from the structure estimation method. Hydrolysis
is not expected to be an important environmental fate process
since this compound lacks functional groups that hydrolyze
under environmental conditions.
Methylglyoxal serves as a substrate for the isozymes E1, E2,
and E3 of human aldehyde dehydrogenase. Oxidation of MG
by these isozymes generated pyruvate. Methylglyoxal is
a partially oxidized compound obtained from the tropospheric
oxidation of numerous hydrocarbons, of both biogenic and
anthropogenic origin. If released to the air, an estimated vapor
pressure of 27 mm Hg at 25 ℃ indicates MG will exist solely as
a vapor in the atmosphere. Vapor-phase MG will be degraded
in the atmosphere by reaction with photochemically produced
hydroxyl radicals; the half-life for this reaction in air is estimated
to be 30 h. Methylglyoxal absorbs light at wavelengths
>290 nm and, therefore, is susceptible to direct photolysis by
sunlight; half-lives of 2–4 h have been reported.
Purification Methods
Commercial 30% (w/v) aqueous solution is diluted to about 10% and distilled twice, taking the fraction boiling below 50o/20mm Hg. (This treatment does not remove lactic acid). [Beilstein 1 IV 3631.]
메틸 글리옥살 준비 용품 및 원자재
원자재
준비 용품