Bivalirudin C화학적 특성, 용도, 생산
개요
Bivalirudin was launched in New Zealand as an anticoagulant for i.v. treatment of
patients with unstable angina undergoing percutaneous transluminal coronary angioplasty.
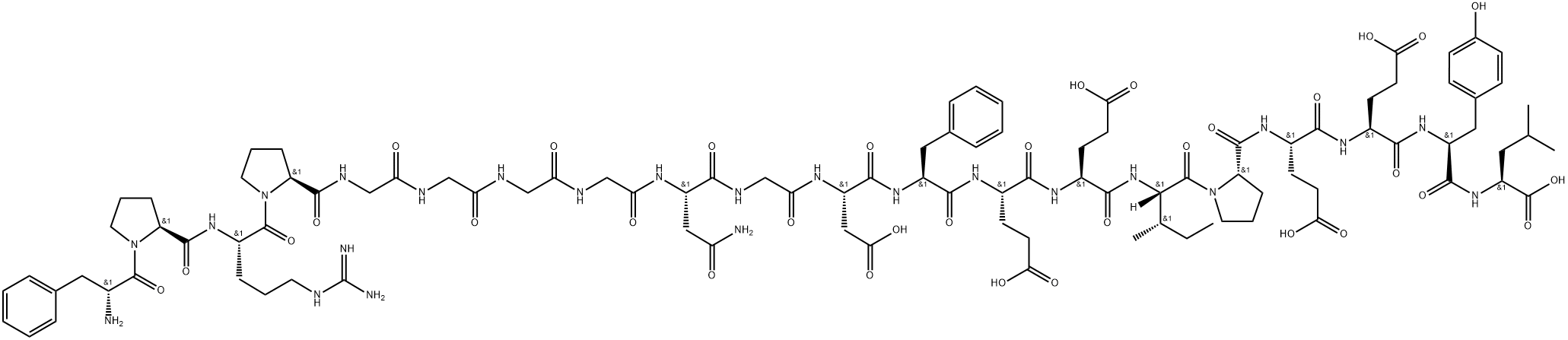
Bivalirubin is a synthetic 20 amino acid peptide rationally modeled on hirudin (residues 53-
64), the most potent and specific naturally-occuring known inhibitor of thrombin, the
enzyme that plays a key role in hemostasis and blood clot formation. This peptide is a
direct thrombin inhibitor that maintains the unique bivalent binding properties of hirudin to
the catalytic site and to the fibrin-recognition exosite of the enzyme, so acting directly on
thrombin with high affinity and specificity. In vitro studies demonstrated that alpha- and
zeta-thrombins, both with the higher fibrinogen-procoagulant activities, were inhibited. In
rats receiving high doses of bivalirudin, the platelet deposition in carotide was reduced by
63% compared to controls. The results of clinical studies, conducted only in patients
receiving concomitant aspirin, suggested that the use of bivalirudin was more efficacious
and more predictable than unfractionated heparin, with fewer bleeding complications.
Despite some unresolved developmental issues, the attractive properties of this novel
agent could make it a useful alternative to heparin in a variety of coagulation disorders.
용도
Anticoagulant; antithrombotic.
정의
ChEBI: A synthetic peptide of 20 amino acids, comprising D-Phe, Pro, Arg, Pro, Gly, Gly, Gly, Gly, Asn, Gly, Asp, Phe, Glu, Glu, Ile, Pro, Glu, Glu, Tyr, and Leu in sequence. A congener of hirudin (a naturally occurring drug found in the saliva o
the medicinal leech), it a specific and reversible inhibitor of thrombin, and is used as an anticoagulant.
Mechanism of action
Bivalirudin is a rapid-onset, short-acting DTI that binds to both the active site and the exosite-1 of
thrombin. Unlike lepirudin, bivalirudin is a reversible inhibitor of both free thrombin and thrombin
bound to fibrin. This reversibility is possible because the bound bivalirudin undergoes cleavage at
the second N-terminal proline to release the portion of the drug bound to the active site. The
carboxyl-terminal portion of bivalirudin dissociates from thrombin to regenerate thrombin. Bivalirudin does not bind to plasma protein.
Pharmacokinetics
Bivalirudin is administered via intravenous bolus injection, followed by continuous infusion (Table
31.4). The drug exhibits a rapid onset and a short duration of action. Bivalirudin is eliminated by
renal excretion. It has been suggested that dosage adjustments be made in patients with severe
renal impairment and in patients undergoing dialysis. Approximately 30% is eliminated unchanged
along with proteolytic cleavage products. Because of the reversible nature of bivalirudin the drug
exhibits less risk of bleeding than other antithrombotics, and there have been no reported cases of
antibody formation to bivalirudin.
Clinical Use
Bivalirudin, a 20-amino-acid peptide, has been approved for use in patients with unstable angina
undergoing percutaneous coronary intervention.
Bivalirudin 준비 용품 및 원자재
원자재
준비 용품