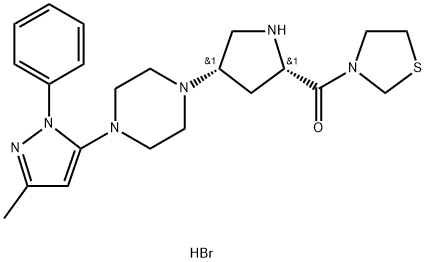
Teneligliptin Hydrobromide
- CAS No.
- 906093-29-6
- Chemical Name:
- Teneligliptin Hydrobromide
- Synonyms
- Teneligliptin HydrobroMide Hydrate;Teneligliptin HBr;3-(((2s,4s)-4-(4-(3-Methyl-1-phenyl-1h-pyrazol-5-yl)-1-piperazinyl)-2-pyrrolidinyl)carbonyl)-thiazolidine hydrobromide (2:5) X H2O;API(Teneligliptin hydro bromide hydrate);3-[[(2S,4S)-4-[4-(3-Methyl-1-phenyl-1H-pyrazol-5-yl)-1-piperazinyl]-2-pyrrolidinyl]carbonyl]-thiazolidine hydrobromide (2:5);[(2S,4S)-4-[4-(5-methyl-2-phenylpyrazol-3-yl)piperazin-1-yl]pyrrolidin-2-yl]-(1,3-thiazolidin-3-yl)methanone,pentahydrobromide;MP-513 (hydrobromide);Ticagrelor hydrobromide;Tepagliptin (Hydrobromide);Teneligliptin (hydrobroMide)
- CBNumber:
- CB12666692
- Molecular Formula:
- C22H31BrN6OS
- Molecular Weight:
- 507.5
- MOL File:
- 906093-29-6.mol
- Modify Date:
- 2024/1/2 16:51:54
Teneligliptin Hydrobromide Chemical Properties,Uses,Production
Uses
Teneligliptin Hydrobromide (2:5) is a dipeptidyl peptidase-4 (DPP-4) inhibitor that is used to treat type 2 diabetes. It is eliminated via excretion, and has a half-life of 24.2 hours in the human body.
Clinical Use
Teneligliptin is a DPP-4 inhibitor which was approved in Japan in 2012 for the treatment of type II diabetes. It was discovered and developed by Mitsubishi Tanabe Pharma under the trade name Tenelia®. Similar to other marketed DPP-4 inhibitors, teneligliptin was well tolerated in all studies and QD dosing produced a long-lasting inhibitory action against DPP-4 and an increase in active GLP-1 levels, with very low rates of renal excretion.
Synthesis
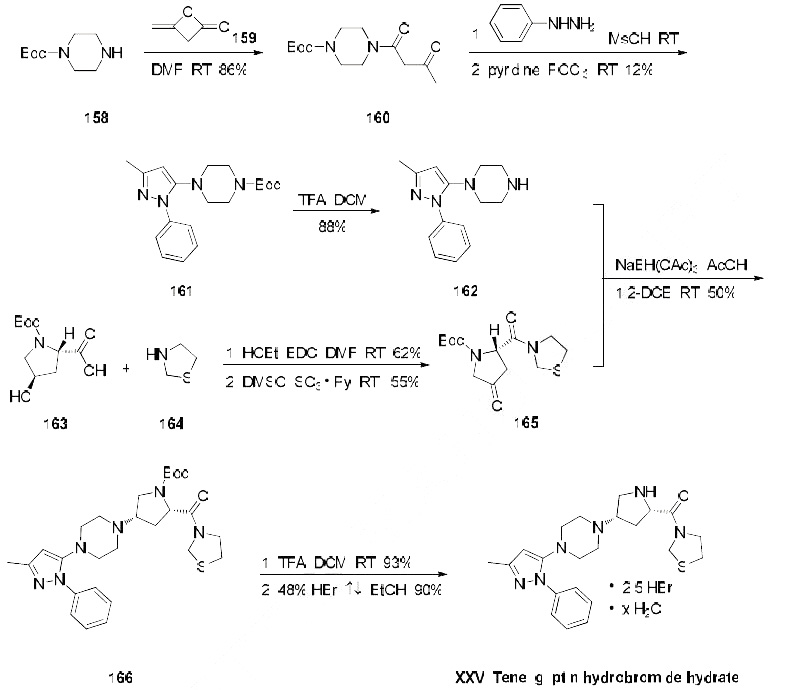
The only reported synthesis of teneligliptin is described in the scheme below. Reaction of commercially available N-Boc-piperazine (158) with diketene (159) in DMF at room temperature gave acetoacetamide 160 in 86% yield, and this material was immediately condensed with phenylhydrazine in methanesulfonic acid followed by a cyclodehydration with phosphorus oxychloride to give pyrazole 161 in 12% yield. The t-butyl carbamate was then removed with TFA in dichloromethane to give amine 162 in 88% yield. This amine was then subjected to butyrolactam 165 (which was prepared from N-Boctrans- 4-hydroxy-L-proline (163) coupled with thiazolidine (164) under conventional amide-forming conditions using EDC) in the presence of sodium triacetoxy borohydride (STAB-H) in acetic acid. This reductive amination reaction afforded the cis-aminopyrrolidine 166 exclusively in 50% yield. Removal of the t-butyl carbamate group with TFA afforded the teneligliptin free amine in 93% yield, and this freebase was then subsequently treated with 48% hydrobromic acid in refluxing ethanol to give teleligliptin hydrobromide hydrate (XXV) in 90% yield.
Teneligliptin Hydrobromide Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| KARPSCHEM LABORATORIES | +91-7249203006 +91-7249203006 | Maharashtra, India | 786 | 58 | Inquiry |
| Metrochem API Private Limited | +91-4069069999 +91-4069069999 | Telangana, India | 78 | 58 | Inquiry |
| Aalidhra Pharmachem Pvt Ltd | +91-9879180894 +91-9879180894 | Gujarat, India | 38 | 58 | Inquiry |
| Lee Pharma Ltd | +91-9849085929 +91-9849085929 | Hyderabad, India | 42 | 58 | Inquiry |
| Soujanya Life Sciences | +91-9717304919 +91-9717304919 | Mumbai, India | 17 | 58 | Inquiry |
| Ami Lifesciences Pvt Ltd | +91-9426998100 +91-9426998100 | Gujarat, India | 62 | 58 | Inquiry |
| BDR Pharmaceuticals International Pvt Ltd | +91-2240560560 +91-7718884418 | Maharashtra, India | 206 | 58 | Inquiry |
| SynZeal Research Pvt Ltd | +1 226-802-2078 | Gujarat, India | 6522 | 58 | Inquiry |
| Pharma Affiliates | 172-5066494 | Haryana, India | 6761 | 58 | Inquiry |
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
906093-29-6(Teneligliptin Hydrobromide)Related Search:
1of4
chevron_right





